- Fact sheets
- Facts in pictures
Publications
- Questions and answers
- Tools and toolkits
- HIV and AIDS
- Hypertension
- Mental disorders
- Top 10 causes of death
- All countries
- Eastern Mediterranean
- South-East Asia
- Western Pacific
- Data by country
- Country presence
- Country strengthening
- Country cooperation strategies
- News releases
- Feature stories
- Press conferences
- Commentaries
- Photo library
- Afghanistan
- Cholera
- Coronavirus disease (COVID-19)
- Greater Horn of Africa
- Israel and occupied Palestinian territory
- Disease Outbreak News
- Situation reports
- Weekly Epidemiological Record
- Surveillance
- Health emergency appeal
- International Health Regulations
- Independent Oversight and Advisory Committee
- Classifications
- Data collections
- Global Health Estimates
- Mortality Database
- Sustainable Development Goals
- Health Inequality Monitor
- Triple Billion
- Data collection tools
- Global Health Observatory
- Insights and visualizations
- COVID excess deaths
- World Health Statistics
- Partnerships
- Committees and advisory groups
- Collaborating centres
- Technical teams
- Organizational structure
- Initiatives
- General Programme of Work
- WHO Academy
- Investment case
- WHO Foundation
- External audit
- Financial statements
- Internal audit and investigations
- Programme Budget
- Results reports
- Governing bodies
- World Health Assembly
- Executive Board
- Member States Portal
Areas of work
SRH and HRP work collaboratively with partners across the world to shape global thinking on sexual and reproductive health by providing new ideas and insights.
We work together to enable high-impact research, inform WHO norms and standards, support research capacity strengthening in low- and middle-income settings, and facilitate the uptake of innovations and new information – including through digital technologies and the research and development of new medicines and devices. An ethical, human rights–based approach that aims to reduce gender inequalities is integrated throughout our work.
We strive for a world where human rights that enable sexual and reproductive health are safeguarded, and where all people have access to quality and affordable sexual and reproductive health information and services.


Working for a brighter, healthier future
WHO has progressively strengthened its work for adolescent health, growing its portfolio of research, norms and standards, country support and advocacy,...

Expert meeting on violence against women and disability
As part of the UN Women-WHO Joint Programme on Strengthening Methodologies and Measurement and Building National Capacities for Violence Against Women Data...

WHO recommendations on self-care interventions: Making over-the-counter emergency contraceptive pills...
WHO strongly recommends making over-the-counter emergency contraceptive pills available without a prescription to individuals who wish to use emergency contraception. This...
Sexual and reproductive health for all: 20 years of the Global Strategy
Special programme on sexual and reproductive health reports on year of progress
Challenging harmful masculinities and engaging men and boys in sexual and reproductive health

- Feature Article
- Developmental Biology
Meta-Research: The need for more research into reproductive health and disease
- Natalie D Mercuri
- Department of Physiology, University of Toronto, Canada ;
- Open access
- Copyright information
- Comment Open annotations (there are currently 0 annotations on this page).
- 2,443 views
- 159 downloads
- 5 citations
Share this article
Cite this article.
- Brian J Cox
- Copy to clipboard
- Download BibTeX
- Download .RIS
- Figures and data
Introduction
Conclusions, data availability, decision letter, author response, article and author information.
Reproductive diseases have a significant impact on human health, especially on women’s health: endometriosis affects 10% of all reproductive-aged women but is often undiagnosed for many years, and preeclampsia claims over 70,000 maternal and 500,000 neonatal lives every year. Infertility rates are also rising. However, relatively few new treatments or diagnostics for reproductive diseases have emerged in recent decades. Here, based on analyses of PubMed, we report that the number of research articles published on non-reproductive organs is 4.5 times higher than the number published on reproductive organs. Moreover, for the two most-researched reproductive organs (breast and prostate), the focus is on non-reproductive diseases such as cancer. Further, analyses of grant databases maintained by the Canadian Institutes of Health Research and the National Institutes of Health in the United States show that the number of grants for research on non-reproductive organs is 6–7 times higher than the number for reproductive organs. Our results suggest that there are too few researchers working in the field of reproductive health and disease, and that funders, educators and the research community must take action to combat this longstanding disregard for reproductive science.
It is difficult to overstate the impact of reproductive disease. Adverse pregnancy outcomes – which include preterm delivery, low birth weight, hypertensive disorders, and gestational diabetes –impact the acute and chronic health of the population ( Barker, 1997 ; Williams, 2011 ; Lewis et al., 2012 ). About 20% of all pregnancies require medical intervention ( Murray and Lopez, 1998 ), and in lower resource settings, pregnancy and delivery complications are a leading cause of maternal and neonatal death ( WHO, 2019 ).
In 1992, the Institute of Medicine in the United States published a report called Strengthening Research in Academic OB-GYN Departments that outlined areas of research with obstetrics and gynecology where improvements were needed, such as low-birth-weight infants, fertility complications, and pregnancy-induced hypertension ( Institute of Medicine, 1992 ). Three decades later, despite the essential nature and impact of the reproductive system, these issues are still major challenges in reproductive health.
Gender inequality and bias have been issues since the onset of biological and medical research. For example, including women as subjects in clinical research was not standard practice until after 1986 ( Liu and Mager, 2016 ). There has been progress in developing policies to increase the representation of women (as both subjects and researchers) and in providing education on gender inequality for all researchers, but women are still underrepresented in scientific and medical research ( Huang et al., 2020 ).
There are a variety of stigmas and taboos surrounding any topic relating to reproductive function. Menstruation is one function that has faced stigmatization that persists today ( Litman, 2018 ; Pickering, 2019 ), with women often feeling too embarrassed to talk about this natural process or even complete an essential task, such as purchasing menstrual products at a local store. Political power highly affects reproductive health care and rights over other biological processes. In many countries, ongoing political and legal battles directly affect access to safe reproductive health care, including contraception, safe abortion, and gender identity rights ( Pugh, 2019 ). There are parallels between the low level of research into reproductive diseases and the response to the AIDS epidemic in the 1980s. The long delay in recognizing AIDS as a significant health issue, and then implementing research policies, perpetuated false ideas surrounding the lifestyles of those affected by the disease and created a barrier to expanding sexual education and seeking healthcare, likely costing many lives ( Francis, 2012 ). Despite great advances in AIDS research and treatment, including social awareness, public health stigma still lingers in society ( Turan et al., 2017 ). Similar increases in advocacy and public awareness are needed to overcome these barriers affecting reproductive health.
Reproductive pathologies are often challenging to diagnose and properly treat, which increases the risk of comorbidity development. Moreover, a long-standing lack of research into reproductive health and disease means that the acute and chronic healthcare burden caused by reproductive pathologies is likely to continue increasing. This lack of research likely results from historic and ongoing systemic biases against female-focused research, and from political and legal challenges to female reproductive health ( Coen-Sanchez et al., 2022 ). In this exploratory analysis we seek to understand the “research gap” between reproductive health and disease and other areas of medical research, and to suggest ways of closing this gap.
Comparing numbers of publications
To benchmark research on reproductive health and disease, we used the PubMed database to compare the number of articles published on seven reproductive organs and seven non-reproductive organs between 1966 and 2021 ( Table 1 ). While the reproductive organs are not essential to postnatal life, we posit that the placenta and the uterus are as essential to fetal survival in utero as the lungs and the heart are to postnatal survival after birth. Our analysis revealed that the average number of articles on non-reproductive organs was 4.5 times higher than the number on reproductive organs (and ranged between about 2 and 20 in pairwise comparisons). The reproductive organs with the most publications were the breast and prostate.
Total number of matching articles from PubMed for seven non-reproductive keywords and seven reproductive keywords for the period 1966–2021.
| Keyword | Total matching articles |
|---|---|
| Non-reproductive keywords | |
| 1,058,995 | |
| 851,955 | |
| 834,006 | |
| 652,797 | |
| 451,177 | |
| 120,034 | |
| 99,772 | |
| Reproductive keywords | |
| 464,629 | |
| 197,736 | |
| 83,971 | |
| 57,076 | |
| 55,971 | |
| 32,344 | |
| 15,019 | |
The research landscape can change over time and efforts to reduce gender bias in research might have had an impact on the volume of reproductive research, so we plotted the number of publications on the 14 organs as a function year between 1966 and 2021 ( Figure 1A ). Breast and prostate were the only reproductive organs to increase in publication at a rate similar to the kidney; the second least studied non-reproductive organ in our list. The intestine was the only non-reproductive organ to show similar publication rates to the other five reproductive organs. To investigate further, we compared disease-driven research versus research not related to disease.
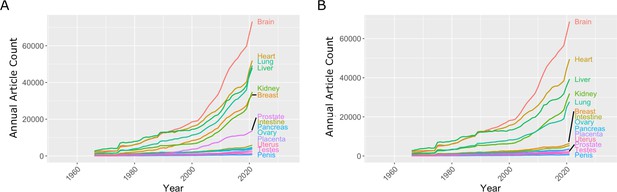
Number of articles published every year on seven reproductive organs and seven non-reproductive organs.
( A ) The number of articles published on most of the non-reproductive organs (including the brain, heart, lung and liver) has increased more rapidly than the number of articles published on the reproductive organs. ( B ) Removing articles that contain the keyword cancer has relatively little effect on the number of articles for non-reproductive organs (with the exception of the lung), but has a significant impact on the number of articles for the two reproductive organs with the most articles: the breast and prostate. Data extracted from PubMed using organ-specific keyword searches for the period 1966–2021.
Figure 1—source data 1
Articles per year for reproductive and non-reproductive organs, with and without the keyword cancer.
Comparing research related to disease and research not related to disease
In the 1970s, the National Institutes of Health (NIH) initiated a war on cancer, and the breast and prostate are both associated with sex-specific cancers. We reassessed publication data with the added search parameter "NOT cancer" to eliminate cancer-based research ( Figure 1B ). We observed a reduction of approximately 20% for most non-reproductive organs; however, the reduction for publication on the breast and prostate was about 80%, suggesting that most research on these organs is driven by an interest in cancer research rather than reproductive health and disease ( Figure 1B ).
Then, for each organ, we plotted the number of publications related to disease on the vertical axis, and the number not related to disease on the horizontal axis, which revealed a high degree of variation among the organs ( Figure 2 ). For three non-reproductive organs (brain, heart, and liver) the number of publications not related to disease was almost three times as high as the number related to disease, and for two non-reproductive organs (kidney and lung) the numbers were similar. For the breast and prostate, on the other hand, the number of publications related to disease was three times as high as the number not related to disease. For the five remaining reproductive organs, and also for the intestine and pancreas, the number of publications not related to disease was about twice as high as the number related to disease (although the total number of publications for these seven organs was about an order of magnitude lower than the number for the other seven organs).
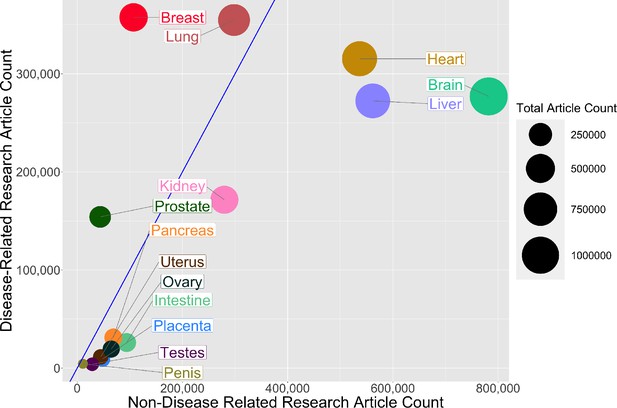
Comparing research related to disease and research not related to disease for reproductive and non-reproductive organs.
For each organ (colored circles) the vertical axis shows the number of publications for the period 1966–2021 related to disease, and the horizontal axis shows the number not related to disease: the area of the circle is proportional to the total number of publications. The straight blue line corresponds to equal numbers of disease-related and non-disease-related publications, so organs to the right of this line (notably non-reproductive organs such as the brain, heart and liver) tend to be the subject of more basic or non-disease-related research, whereas organs to the left of this line (notably reproductive organs such as the breast and prostate) tend to be the subject of disease-related research. The lung is the only non-reproductive organ in our sample to the left of the blue line.
Figure 2—source data 1
Total number of articles on research related to disease and research not related to disease for reproductive and non-reproductive organs.
Research funding
Next we used databases belonging to the Canadian Institutes of Health Research (CIHR) and the NIH to investigate funding trends for the different organs. The 14 keywords (brain, heart, liver, lung, kidney, intestine, pancreas, breast, prostate, ovary, uterus, penis, testes, and placenta) were entered into each database, and we extracted funding data for the period between 2013 and 2018. These organs were chosen as keywords to investigate the funding related to a basic understanding of the biology of these organs. Although grants that relate to pregnancy or fertility may not be captured, these topics are much broader and would introduce subtopics outside of the reproductive scope, similar to using keywords such as metabolism or behaviour. Table 2 gives the number of projects for each keyword and the corresponding average funding amount per grant for the CIHR, and the same for the NIH. Our analysis found that the mean grant amounts for the CIHR and NIH are similar between different keyword research topics (CIHR: $ 370 000 ± $ 50 000; NIH: $ 481 500 ± $ 50 000). The similar funding amounts between different organs are encouraging and may result from standard funding guidelines for biomedical research. However, our analysis found that the average number of funded projects is much higher for non-reproductive organs compared to reproductive organs for both the CIHR (800 vs 115) and the NIH (31 000 vs 5 300).
Total number of projects funded and average grant (in Canadian or US dollars) for the Canadian Institutes of Health Research (columns 2 and 3) and the US National Institutes of Health (columns 4 and 5) for the years 2013–2018 for seven non-reproductive keywords and seven reproductive keywords (column 1).
| Keyword | Number of projects (CIHR) | Average grant funded (CAD) | Number of projects(NIH) | Average grant funded(USD) |
|---|---|---|---|---|
| Non-reproductive keywords | ||||
| 1686 | $391,023 | 81666 | $441,149 | |
| 1214 | $369,665 | 43833 | $491,993 | |
| 1597 | $314,473 | 22072 | $454,276 | |
| 526 | $371,154 | 34492 | $525,631 | |
| 347 | $424,360 | 21176 | $508,853 | |
| 128 | $444,490 | 5800 | $371,727 | |
| 96 | $491,274 | 8649 | $482,901 | |
| Reproductive keywords | ||||
| 459 | $336,734 | 19132 | $525,134 | |
| 143 | $299,034 | 8960 | $514,638 | |
| 42 | $379,349 | 4814 | $520,804 | |
| 105 | $369,825 | 2169 | $526,147 | |
| 45 | $324,690 | 1356 | $509,250 | |
| 10 | $372,110 | 340 | $500,160 | |
| 1 | $304,676 | 323 | $369,434 | |
Table 2—source data 1
Source data for Table 2 .
Our analysis suggests a bias against research into reproductive health and disease, and it is important that efforts are made to eliminate this bias so that research into reproductive medicine does not fall further behind. The higher levels of research observed for some reproductive organs (notably the breast and prostate) were driven by cancer-focused research, but this has not led to an increase in the level of non-disease-related research on these organs ( Figure 1B ). Factors such as Breast Cancer Awareness Month ( Jacobsen and Jacobsen, 2011 ) and screening programmes for prostate cancer ( Dickinson et al., 2016 ) likely led to the increase in publications about these two reproductive organs.
While our analysis is suggestive that many reproductive organs achieve a good balance of non-disease versus disease-related research, the paucity of research is highly problematic to the field. An important consideration is that a lack of non-disease-related research on reproductive organs may hinder progress in diagnosing and treating a wide range of pathologies (including preeclampsia, polycystic ovary syndrome, and endometriosis).
In a competitive funding system, publications are correlated to successful grants and dollar values awarded. Across research areas, we found that the mean grant dollar amounts per project are similar. However, the numbers of funded research projects on non-reproductive organs were higher than the numbers for reproductive organs by a factor of 6–7 (which is slightly larger than the discrepancy seen in publication rates). An important consideration is that the part of the NIH that supports reproductive research in the US, the National Institute of Child Health and Development, is one of the lowest-funded institutes at the NIH and does not have the word reproduction in its title. In Canada, the Human Development, Child and Youth Health Institute of CIHR is a funder of most pregnancy and reproductive biology grants, typically awarded through the Clinical Investigation – A panel, and it may be that the use of a clinical panel to fund this area of research inhibits non-diseased focused research. This panel is well-funded relative to other panels; however, some research areas (e.g., cardiovascular and neurological research) have more than one panel.
A growing political and societal emphasis is placed on disease-related research, such as cancer. This may arise from a view of basic research as ineffective or inefficient compared to applied research ( Lee, 2019 ). Perhaps this is best seen in our analysis by the high percentage of research publications on the prostate and breast that are due to cancer research, whereas most research on the other reproductive organs we studied was not disease-related. While the placenta and uterus are widely viewed as causal organs for reproductive complications that claim large numbers of maternal and neonatal lives, and treatments cost tens of billions of US dollars every year, there is relatively little disease-related research into these organs. The investigation of cancer biology within a reproductive organ can rely on knowledge of cancer in other organ systems. However, the low levels of research into reproductive organs relative to other organs means that there is much less foundational knowledge to rely on when seeking to develop treatments for diseases of these organs. Moreover, there are fewer researchers who are experienced on working with these organs.
There are several limitations to our approach. One important limitation is that the number of unfunded grant applications is not accessible, so we could not determine if the lower numbers of grants for research on reproductive health and disease were due to proportionally lower total application numbers, or to a bias against reproductive research. Funding bodies should conduct internal analyses to determine appropriate action. The use of keywords to distinguish between non-disease and disease-related research is a limitation, and the relatively low numbers of publications on reproductive organs can also present challenges when making comparisons. However, the differences we observe between research into reproductive and non-reproductive organs (as measured by numbers of publications and levels of funding) are large and are unlikely to result from missing search terms.
How can we address the research gap and enable the field of reproductive health and disease to catch up with other areas of research? Based on our analysis, we need to increase the number of researchers working on reproductive organs and related pathologies. Recent efforts by the NIH, such as the Human Placenta Project ( Guttmacher et al., 2014 ), indicate a recognition of the need to increase research capacity in reproductive sciences, and may lead to further increases in both interest and research capacity in the longer term.
New researchers may avoid the reproductive field due to social and political factors and the research gap (ie, the low levels of grant funding and publications), and this in turn may discourage students and trainees, which will make it even more difficult to increase the size of the research base. While continued advocacy, education, and political lobbying may help to overcome many of the social and political factors, closing the research gap will require other approaches.
To increase researchers and research output, we may learn lessons from the examples of breast and prostate cancer. In both cases, research increased dramatically from a historically low level. While public campaigns played a prominent role in these increases, the existence of a large pool of researchers and trainees already working on other types of cancers was probably more important (as it was these researchers, rather than those doing non-disease-related research on these organs, who did most of the work on breast and prostate cancer). However, this is unlikely to work for preeclampsia and other reproductive pathologies as there are no large pools of existing researchers available to switch the focus of their work.
Therefore, to increase research capacity, we should promote collaborations between researchers working on reproductive health and disease and those working in other areas of physiology and medicine, especially other areas with much higher research capacities. There are plenty of examples that show the benefit of such an integrated approach. For instance, female sex hormones protect against many aging diseases, such as cardiovascular and neurological diseases, leading to the prescription of hormone replacement therapies after menopause in some women ( Paciuc, 2020 ).
Links to immunology, cardiology and other systems can be used to increase research capacity. During pregnancy, there are dramatic changes in maternal physiology, including metabolism, the immune system, and cardio-pulmonary systems, and consequently, these are the same systems affected by reproductive pathologies. Preeclampsia predisposes the mother to a long-term cardiovascular risk of developing peripheral artery disease, coronary artery disease, and congestive heart failure ( Rana et al., 2019 ). Additionally, complications of the liver and kidney are associated with preeclampsia. Polycystic ovary syndrome and endometriosis are related to metabolism problems and the risk of cancer development. Children born from pregnancies affected by preeclampsia or fetal growth restriction are at a 2.5 times higher risk of developing hypertension and require anti-hypertensive medications as adults ( Ferreira et al., 2009 ; Fox et al., 2019 ).
The pathological interaction of reproductive with non-reproductive systems and organs should attract investigators from nephrology, hepatology and cardiovascular research, where the total number of researchers is 10–20 times as high as the number in reproductive health and disease. If just 1% of the researchers in the cardiovascular field were to refocus on pregnancy-related cardiovascular adaptation and pathologies, this would increase reproductive research by 10%.
Our neglect of the placenta and reproductive biology impedes other biomedical research areas. In cancer research, the methylation patterns of tumours look most like those found in the placenta, but why placenta methylation patterns are so unlike all other organs is not known ( Smith et al., 2017 ; Rousseaux et al., 2013 ). In regenerative medicine, the immune-modulating genes used by the placenta ( Szekeres-Bartho, 2002 ) are repurposed to generate universally transplantable stem cells and tissues ( Han et al., 2019 ). A poor understanding of reproductive biology is dangerous, considering emerging diseases that affect pregnancy and fetal development, such as the recent Zika virus outbreak ( Schuler-Faccini et al., 2016 ; Calvet et al., 2016 ). There are likely many other broad benefits to better understanding reproductive biology. The time to act is now, as waiting longer will not improve the situation.
Publication rates
Published research manuscripts were searched in NCBI’s PubMed database ( https://pubmed.ncbi.nlm.nih.gov/ ) between and including the years 1966 and 2021. Keywords for each search pertained to a specific organ or disease and were limited to the title/abstract of the manuscripts. The organs used for these analyses were the brain, heart, liver, lung, kidney, intestine, pancreas, breast, prostate, ovary, uterus, penis, testes, and placenta. We restricted the organ publication timelines to the years 1966–2021 and extracted the annual article count. The organ publication timeline was reconducted with the addition of the search parameter "NOT cancer".
Funding rates
Grant funding data was obtained from the CIHR funding database ( https://webapps.cihr-irsc.gc.ca/funding/Search?p_language=E&p_version=CIHR ) and the NIH reporter tool ( https://reporter.nih.gov ) by searching keywords in the title and abstracts/summary. Keywords used for these searches were brain, heart, liver, lung, kidney, intestine, pancreas, breast, prostate, ovary, uterus, penis, testes, and placenta. The years were restricted to 2013–2018. The total number of projects pertaining to each search term during this period was extracted, and the total amount of funding for those projects was averaged.
All graphs were produced using R (version 4.0.2) in R Studio (version 1.3.1073). R packages used were ggplot2, tidyverse, formattable, gridExtra, RColorBrewer, ggrepel.
All data were obtained from public databases (PubMed/NCBI, NIH and CIHR). Source data files for Figure 1, Figure 2 and Table 2 are available (see figure and table captions for details).
- Google Scholar
- de Filippis I
- de Sequeira PC
- de Mendonça MCL
- de Oliveira L
- Tschoeke DA
- Thompson FL
- Dos Santos FB
- Nogueira RMR
- de Filippis AMB
- Coen-Sanchez K
- El-Mowafi IM
- Idriss-Wheeler D
- Dickinson J
- Connor Gorber S
- Stehouwer CDA
- Lewandowski AJ
- Guttmacher AE
- Ferreira LMR
- Strominger JL
- Meissner TB
- Barabási AL
- Institute of Medicine
- Jacobsen GD
- Jacobsen KH
- Pickering K
- Karumanchi SA
- Rousseaux S
- Debernardi A
- Nagy-Mignotte H
- Moro-Sibilot D
- Brichon P-Y
- Lantuejoul S
- de Reyniès A
- Brambilla C
- Brambilla E
- Schuler-Faccini L
- Feitosa IML
- Horovitz DDG
- Cavalcanti DP
- Doriqui MJR
- Wanderley HYC
- El-Husny AS
- Sanseverino MTV
- Brazilian Medical Genetics Society–Zika Embryopathy Task Force
- Cacchiarelli D
- Szekeres-Bartho J
- Browning WR
- Mugavero MJ
- Peter Rodgers Senior and Reviewing Editor; eLife, United Kingdom
- Marleen van Gelder Reviewer
- James Roberts Reviewer
In the interests of transparency, eLife publishes the most substantive revision requests and the accompanying author responses.
Decision letter after peer review:
Thank you for submitting the paper "A Poor Research Landscape Hinders the Progression of Knowledge and Treatment of Reproductive Diseases" for consideration by eLife . Your article has been reviewed by 3 peer reviewers, and the evaluation has been overseen by a Reviewing Editor and a Senior Editor. The following individuals involved in review of your submission have agreed to reveal their identity: Marleen van Gelder (Reviewer #1); James Roberts (Reviewer #3).
This article will need considerable revision to be suitable for publication as a Feature Article. In particular, you will need to address the concerns raised by the referees (see below), and also address a number of editorial points.
Reviewer #1
In this manuscript, Mercuri and Cox aimed to quantify the advancement of research in reproductive sciences relative to other medical disciplines. They compared two indicators of the research landscape: published research manuscripts and funded projects. The results showed lower publication rates for research on reproductive organs compared to selected non-reproductive organs, in particular concerning basic research. In addition, a relatively small number of grants was funded for projects on diseases with a reproductive focus. Based on these data, the authors concluded that the gap in knowledge and treatment of diseases of the reproductive organs is at least partially caused by a poor research landscape.
Although the conclusions of this paper are somewhat supported by the data, some aspects of the methods and reporting need to be clarified.
[Note: The following point is covered by the queries in the Word version I have sent you]
1) The manuscript, and in particular the Introduction and Discussion sections, could benefit from restructuring, in which adhering to a relevant reporting guideline may be helpful. For example, the authors provide relatively extensive background information on a number of important reproductive health disorders, but the level of detail does not contribute to setting the aim for the study. Moreover, the last paragraph of the introduction section (lines 92-100) already seems to include the conclusion of this paper.
[Note: Please address points b, d and f below. The other points are covered by the queries in the Word version I have sent you]
2) Concerns regarding the methods:
a) Citations in PubMed are known to be selective before 1966; consider using a fixed start date/year for the search.
b) The results strongly depend on the organs and diseases selected to be included in the 'reference group'. Provide a rationale for the selection of organs, which in the current analysis only seem to include major organs that are known to be well-studied, and not organs such as skin, eyes, intestine, pancreas, spleen or urinary bladder. The selection is vital for drawing robust conclusions from the data.
c) The approach to distinguish between basic and applied research is not validated.
d) The prevalence of diseases reported in Figure 4 is highly country-specific, in particular for tuberculosis. Therefore, this comparison may not be suitable for an international audience.
e) The most important limitation of the grant funding data was already mentioned: "the number and keywords of failed grant applications were not accessible" (lines 271-272). Therefore, it is hard to draw conclusions on failure of grant applications on reproductive health.
f) The rationale for the keywords used in the funding databases is missing and likely to yield selective results. Many reproductive health related projects may be missed, as keywords such as pregnancy and subfertility were not included. And also in this search, the selection of keywords for the reference group seems biased.
[Note: Please consider adding a table as suggested below; however, this is optional rather than essential.]
3) To emphasize the lack of knowledge in relation to disease burden, a table summarizing the prevalence, number of publications, and grants could summarize the results.
[Note: This point is covered by the queries in the Word version I have sent you]
4) A number of topics and statements in the Discussion section seem to be unrelated to the aim of this study. Examples include the female representation in STEM disciplines and the correlation between research publications and changes in policy (this was not specifically analyzed and would require additional analyses).
Reviewer #2
[Note: Please address the following point]
While the authors have attempted to be broad in their assessment of reproduction research, they seem to neglect two very broad areas of women's health for which there is little research: menstruation and menopause. Both are only mentioned in the discussion, and referenced with respect to promotion of the study of human physiology. Given the focus on lack of basic understanding of reproductive organs, it may be worth mentioning these, particularly in comparison to the depth of research on erectile dysfunction; this may also help to emphasize the fact that the lack of research in reproduction primarily affects women (though there are of course consequences for men's health, including the period in the womb).
Figure 1: the color code is not clear; Not sure how this could be better represented, but maybe listing the organs from high to low for both parts a and b in the legend? Or magnifying one part of each graph? In particular, the 80% loss of publications in breast/prostate when applying the search term "NOT cancer" does not come through; so perhaps a graph focusing on just these two organs showing the original search and the "NOT cancer" search results would be best?
Tables 2 and 3: It is not clear how this search was done; was the project title or abstract of grants searched for these key terms?
Discussion (including lines 259-260): I'm not sure that the conclusion drawn here is consistent with the data? The authors somewhat confusingly alternate between lack of research in reproduction as a whole vs. lack of basic research in this area.
[Note: Addressing the following point is optional, not essential.]
Another point of discussion that merits mention here is how the lack of interest/emphasis on reproduction research by funding agencies in turn affects the perception of "impact" of such research: i.e. both in terms of how low impact factors of reproduction journals are compared to journals in other fields, but also how the high-impact journals (Cell/Science/Nature) view/receive submissions from researchers in this area. Reviewer #3
The authors propose that research in reproductive areas lags behind that of other areas of biology. They support this with information from publications and funding sources.
This is a presentation of importance to investigators in all fields, funders and the general public. For reproductive investigators it provides objective data to support the lagging of reproductive research and to investigators in other areas of biology and the general public should be an eye opening demonstration of the huge gap between research in reproduction and other areas of biology. One would hope it would also provide a motivation to funders to modify the situation.
The authors remind us of the importance of reproduction on the survival of the species and provide extensive data on specific examples of the impact of reproductive diseases. They then use review of publications keyed to reproductive organs and non-reproductive organs both currently and over time. They point out that research on non-reproductive organs is 5 to 20 times more frequent than that on reproductive organs. [Note: Please address the point made in the following sentence] They should make it clearer that this is referring to specific organs and not a comparison sum of research on all organs of reproduction and not reproduction. They show that over time this discrepancy has increased with the exception of prostate, and breast research but even with those it is evident this is research related specifically to cancer and not normal organ function.
They make a slightly less compelling comparison on the portion of research devoted to basic understanding or clinical research which for nonreproductive organs is considerably more for basic science than in reproductive organs. [Note: Please address the point made in the following sentence] However, this is likely compromised by the relative minute number of either type of studies in reproduction.
They then make comparisons between the impact of specific reproductive topics and publications. They state that although preeclampsia and breast cancer have a similar prevalence the number of breast cancer publications are much higher. [Note: Please address the point made in the following sentence] To me the comparison of a disorder with high mortality (breast cancer) and far lower mortality (preeclampsia) does not provide a compelling argument and also is a little off target for comparing reproductive and nonreproductive research.
[Note: Please address the point made in the following paragraph]
They make a similar comparison of PCOS a reproductive disorder with other non-reproductive disorders of similar or lower prevalence, autism, tuberculosis, Crohn's Disease and Lupus with a much lower publication rate for PCOS. Again, this seems a bit of comparing apples and oranges.
They investigate the relative funding of research on these topics in the US and Canada and find that the size of individual grants for reproductive and non-reproductive research in both countries is similar but that the number of funded grants for specific non-reproductive organs is, that like that of publications, is about 2 to 20 times higher for nonreproductive organs.
The authors present their conclusions of the reason for the discrepancy. They point out gender bias which has been a target for improvement for several years and has been reduced but research is still not on an equal basis for men and women. However, the bias goes beyond gender since male reproductive research publications and funding also lags. They conclude that there is a general bias against reproductive research. [Note: Please consider mentioning the following point in your article] Interestingly they do not cite a major support for this conclusion, that the major NIH institute supporting reproductive research, the National Institute of Child Health and Development (NICHD)is one lowest funded institutes and does not have reproduction in its title.
They provide two general suggestions to increase reproductive research. The first is to increase funding and the second to involve other forms of research in studies supporting the role of reproductive disorders and physiology in non-reproductive studies. [Note: Please address the point made in the rest of this paragraph] They point out the relationship of preeclampsia to later life cardiovascular disease as an example of this. Unfortunately, they state this relationship as causal which has not been established. Nonetheless studying preeclampsia will likely provide information useful to cardiovascular health.
It is possible that linking publications and funding amounts to conclusions about bias against reproductive research is not precise. However, the magnitude of the differences strongly supports the authors' premise.
This interesting presentation makes and important point about the fact that reproductive research lags beyond other biological research. They do this through the use of publication and grant funding reviews. The differences are large in a direction that support the point they are making. There are some suggestions that I believe would improve the presentation.
[Note: Please address the following three points]
1. There should be a bit more discussion of the limitations of their approach.
2. In the comparisons of disorders of reproduction and non-reproduction they should indicate the limitations of comparing very different disorders.
3. Preeclampsia as a cause of later life CVD has not been established. They are related.
Reviewer #1 In this manuscript, Mercuri and Cox aimed to quantify the advancement of research in reproductive sciences relative to other medical disciplines. They compared two indicators of the research landscape: published research manuscripts and funded projects. The results showed lower publication rates for research on reproductive organs compared to selected non-reproductive organs, in particular concerning basic research. In addition, a relatively small number of grants was funded for projects on diseases with a reproductive focus. Based on these data, the authors concluded that the gap in knowledge and treatment of diseases of the reproductive organs is at least partially caused by a poor research landscape. Although the conclusions of this paper are somewhat supported by the data, some aspects of the methods and reporting need to be clarified. [Note: The following point is covered by the queries in the Word version I have sent you] 1) The manuscript, and in particular the Introduction and Discussion sections, could benefit from restructuring, in which adhering to a relevant reporting guideline may be helpful. For example, the authors provide relatively extensive background information on a number of important reproductive health disorders, but the level of detail does not contribute to setting the aim for the study. Moreover, the last paragraph of the introduction section (lines 92-100) already seems to include the conclusion of this paper.
This query has been responded to the in Word file
[Note: Please address points b, d and f below. The other points are covered by the queries in the Word version I have sent you] 2) Concerns regarding the methods: a) Citations in PubMed are known to be selective before 1966; consider using a fixed start date/year for the search.
This query has been responded to the in Word file. We have now used a fixed date of 1966 as the early timepoint and as indicated in the Word file.
Organs such as brain, heart and lungs are essential for life. The placenta is similarly essential. Other organs such as kidney and liver are also essential but not as immediate. We now include the intestine as a reference point.
Our preliminary analysis found that Skin has over 800,000 publication mentions, but it is not clear if this is the skin organ or a skin on something more work to eliminate background skin hits would be needed. Epidermis has 60,000 hits that are likely more specific, but we did find may abstracts and titles on the skin organ that do not use epidermis. Eyes are nearly 700,000 publications, intestine also over 700,000, pancreas has over 200,000 spleen is also over 200,000 urinary bladder has 130,000, which is similar to the placenta at just over 100,000
This preliminary search seems to still support our conclusion that placenta and reproductive organs are under-researched and only add a list of other organs that are better studied.
Comparisons of diseases has been removed from the manuscript.
We have removed disease focused terms form the search to ensure we capture organ focus research. The inclusion of pregnancy or subfertility would be misleading as it would include disciplines such as sociology and psychology. This is akin to searching for diabetes or metabolism to understand the research landscape on the pancreas.
We felt the separate tables made the information more digestible.
This query has been responded to the in Word file. We have extensively edited and redrafted the Discussion section.
Reviewer #2 [Note: Please address the following point] While the authors have attempted to be broad in their assessment of reproduction research, they seem to neglect two very broad areas of women's health for which there is little research: menstruation and menopause. Both are only mentioned in the discussion, and referenced with respect to promotion of the study of human physiology. Given the focus on lack of basic understanding of reproductive organs, it may be worth mentioning these, particularly in comparison to the depth of research on erectile dysfunction; this may also help to emphasize the fact that the lack of research in reproduction primarily affects women (though there are of course consequences for men's health, including the period in the womb). Figure 1: the color code is not clear; Not sure how this could be better represented, but maybe listing the organs from high to low for both parts a and b in the legend? Or magnifying one part of each graph? In particular, the 80% loss of publications in breast/prostate when applying the search term "NOT cancer" does not come through; so perhaps a graph focusing on just these two organs showing the original search and the "NOT cancer" search results would be best?
These corrections have been made to the in Word file.
We agree and have focused the discussion on the general low level of publications and low level of researchers in the field.
Another point of discussion that merits mention here is how the lack of interest/emphasis on reproduction research by funding agencies in turn affects the perception of "impact" of such research: i.e. both in terms of how low impact factors of reproduction journals are compared to journals in other fields, but also how the high-impact journals (Cell/Science/Nature) view/receive submissions from researchers in this area.
This is an issue many discipline struggle with. A low number of researchers in a field tends to create low levels of impact as measured through citations. Attempts to normalize impact factors and citation rates to the size of the field may help. While we agree with the reviewers comments we cannot address within our study.
Reviewer #3 The authors propose that research in reproductive areas lags behind that of other areas of biology. They support this with information from publications and funding sources. This is a presentation of importance to investigators in all fields, funders and the general public. For reproductive investigators it provides objective data to support the lagging of reproductive research and to investigators in other areas of biology and the general public should be an eye opening demonstration of the huge gap between research in reproduction and other areas of biology. One would hope it would also provide a motivation to funders to modify the situation. The authors remind us of the importance of reproduction on the survival of the species and provide extensive data on specific examples of the impact of reproductive diseases. They then use review of publications keyed to reproductive organs and non-reproductive organs both currently and over time. They point out that research on non-reproductive organs is 5 to 20 times more frequent than that on reproductive organs. [Note: Please address the point made in the following sentence] They should make it clearer that this is referring to specific organs and not a comparison sum of research on all organs of reproduction and not reproduction. They show that over time this discrepancy has increased with the exception of prostate, and breast research but even with those it is evident this is research related specifically to cancer and not normal organ function.
Thank you for this comment. These clarifications have been made to the in Word file.
We agree that the lower level make estimating the ratio of basic to applied very challenging. But there seems to be a tendency to bias to basic research. We made some changes to the results and discussion to acknowledge this challenge.
We agree and have remove the section discussing a comparison of disease prevalence and mortalities. We realize there was no benefit to comparison disease prevalence and severity.
They investigate the relative funding of research on these topics in the US and Canada and find that the size of individual grants for reproductive and non-reproductive research in both countries is similar but that the number of funded grants for specific non-reproductive organs is, that like that of publications, is about 2 to 20 times higher for nonreproductive organs. The authors present their conclusions of the reason for the discrepancy. They point out gender bias which has been a target for improvement for several years and has been reduced but research is still not on an equal basis for men and women. However, the bias goes beyond gender since male reproductive research publications and funding also lags. They conclude that there is a general bias against reproductive research. [Note: Please consider mentioning the following point in your article] Interestingly they do not cite a major support for this conclusion, that the major NIH institute supporting reproductive research, the National Institute of Child Health and Development (NICHD)is one lowest funded institutes and does not have reproduction in its title.
Thank you for this comment, we have added it!
Thank you for the comment, we modified our statement to an observed increased risk of cardiovascular disease, as the risk may be causal or associated as the reviewer stated.
It is possible that linking publications and funding amounts to conclusions about bias against reproductive research is not precise. However, the magnitude of the differences strongly supports the authors' premise. This interesting presentation makes and important point about the fact that reproductive research lags beyond other biological research. They do this through the use of publication and grant funding reviews. The differences are large in a direction that support the point they are making. There are some suggestions that I believe would improve the presentation. 1. There should be a bit more discussion of the limitations of their approach.
We have added more caveats about our approach and interpretation
The comparisons of diseases has been removed.
This is addressed as per the above comment.
Author details
Natalie D Mercuri is in the Department of Physiology, University of Toronto, Toronto, Canada
Contribution
Competing interests.
Brian J Cox is in the Department of Physiology and the Department of Obstetrics and Gynaecology, University of Toronto, Toronto, Canada
For correspondence
University of toronto, canada research chairs.
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
We thank the University of Toronto and the Department of Physiology for providing the opportunity and supporting the completion of this review. We also thank the librarians who offered expert advice on keyword searches of databases.
Publication history
- Received: October 28, 2021
- Preprint posted: November 19, 2021 (view preprint)
- Accepted: December 12, 2022
- Accepted Manuscript published: December 13, 2022 (version 1)
- Accepted Manuscript updated: December 13, 2022 (version 2)
- Version of Record published: December 21, 2022 (version 3)
© 2022, Mercuri and Cox
This article is distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use and redistribution provided that the original author and source are credited.
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as pdf).
- Article PDF
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools), categories and tags.
- reproductive biology
- reproductive health
- meta-research
Research organism
- Of interest
Scaling between cell cycle duration and wing growth is regulated by Fat-Dachsous signaling in Drosophila
Further reading.
The atypical cadherins Fat and Dachsous (Ds) signal through the Hippo pathway to regulate growth of numerous organs, including the Drosophila wing. Here, we find that Ds-Fat signaling tunes a unique feature of cell proliferation found to control the rate of wing growth during the third instar larval phase. The duration of the cell cycle increases in direct proportion to the size of the wing, leading to linear-like growth during the third instar. Ds-Fat signaling enhances the rate at which the cell cycle lengthens with wing size, thus diminishing the rate of wing growth. We show that this results in a complex but stereotyped relative scaling of wing growth with body growth in Drosophila . Finally, we examine the dynamics of Fat and Ds protein distribution in the wing, observing graded distributions that change during growth. However, the significance of these dynamics is unclear since perturbations in expression have negligible impact on wing growth.
- Stem Cells and Regenerative Medicine
Endothelial cell signature in muscle stem cells validated by VEGFA-FLT1-AKT1 axis promoting survival of muscle stem cell
Endothelial and skeletal muscle lineages arise from common embryonic progenitors. Despite their shared developmental origin, adult endothelial cells (ECs) and muscle stem cells (MuSCs) (satellite cells) have been thought to possess distinct gene signatures and signaling pathways. Here we shift this paradigm by uncovering how adult MuSC behavior is affected by the expression of a subset of EC transcripts. We used several computational analyses including single-cell RNAseq to show that MuSCs express low levels of canonical EC markers in mice. We demonstrate that MuSC survival is regulated by one such prototypic endothelial signaling pathway (VEGFA-FLT1). Using pharmacological and genetic gain- and loss-of-function studies, we identify the FLT1-AKT1 axis as the key effector underlying VEGFA-mediated regulation of MuSC survival. All together, our data support that the VEGFA-FLT1-AKT1 pathway promotes MuSC survival during muscle regeneration, and highlights how the minor expression of select transcripts is sufficient for affecting cell behavior.
Loss of ninein interferes with osteoclast formation and causes premature ossification
Ninein is a centrosome protein that has been implicated in microtubule anchorage and centrosome cohesion. Mutations in the human NINEIN gene have been linked to Seckel syndrome and to a rare form of skeletal dysplasia. However, the role of ninein in skeletal development remains unknown. Here, we describe a ninein knockout mouse with advanced endochondral ossification during embryonic development. Although the long bones maintain a regular size, the absence of ninein delays the formation of the bone marrow cavity in the prenatal tibia. Likewise, intramembranous ossification in the skull is more developed, leading to a premature closure of the interfrontal suture. We demonstrate that ninein is strongly expressed in osteoclasts of control mice, and that its absence reduces the fusion of precursor cells into syncytial osteoclasts, whereas the number of osteoblasts remains unaffected. As a consequence, ninein-deficient osteoclasts have a reduced capacity to resorb bone. At the cellular level, the absence of ninein interferes with centrosomal microtubule organization, reduces centrosome cohesion, and provokes the loss of centrosome clustering in multinucleated mature osteoclasts. We propose that centrosomal ninein is important for osteoclast fusion, to enable a functional balance between bone-forming osteoblasts and bone-resorbing osteoclasts during skeletal development.
Be the first to read new articles from eLife

Click through the PLOS taxonomy to find articles in your field.
For more information about PLOS Subject Areas, click here .
Loading metrics
Open Access
Peer-reviewed
Research Article
Evaluating the effectiveness of sexual and reproductive health services during humanitarian crises: A systematic review
Roles Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Writing – original draft, Writing – review & editing
* E-mail: [email protected]
Affiliations Health in Humanitarian Crises Centre, London School of Hygiene & Tropical Medicine, London, United Kingdom, Centre for Maternal, Adolescent, Reproductive and Child Health (MARCH), London School of Hygiene & Tropical Medicine, London, United Kingdom
Roles Formal analysis, Writing – original draft, Writing – review & editing
Affiliation Health in Humanitarian Crises Centre, London School of Hygiene & Tropical Medicine, London, United Kingdom
Roles Conceptualization, Formal analysis, Writing – review & editing
Affiliation Centre for Maternal, Adolescent, Reproductive and Child Health (MARCH), London School of Hygiene & Tropical Medicine, London, United Kingdom
Roles Conceptualization, Funding acquisition, Methodology, Writing – review & editing
Affiliation Department of Reproductive Health and Research, World Health Organization, Geneva, Switzerland
Roles Conceptualization, Funding acquisition, Writing – review & editing
- Neha S. Singh,
- James Smith,
- Sarindi Aryasinghe,
- Rajat Khosla,
- Lale Say,
- Karl Blanchet

- Published: July 6, 2018
- https://doi.org/10.1371/journal.pone.0199300
- Reader Comments
An estimated 32 million women and girls of reproductive age living in emergency situations, all of whom require sexual and reproductive health (SRH) information and services. This systematic review assessed the effect of SRH interventions, including the Minimum Initial Service Package (MISP) on a range of health outcomes from the onset of emergencies.
Methods and findings
We searched EMBASE, Global Health, MEDLINE and PsychINFO databases from January 1, 1980 to April 10, 2017. This review was registered with the PROSPERO database with identifier number CRD42017082102. We found 29 studies meet the inclusion criteria. We found high quality evidence to support the effectiveness of specific SRH interventions, such as home visits and peer-led educational and counselling, training of lower-level health care providers, community health workers (CHWs) to promote SRH services, a three-tiered network of health workers providing reproductive and maternal health services, integration of HIV and SRH services, and men’s discussion groups for reducing intimate partner violence. We found moderate quality evidence to support transport-based referral systems, community-based SRH education, CHW delivery of injectable contraceptives, wider literacy programmes, and birth preparedness interventions. No studies reported interventions related to fistulae, and only one study focused on abortion services.
Conclusions
Despite increased attention to SRH in humanitarian crises, the sector has made little progress in advancing the evidence base for the effectiveness of SRH interventions, including the MISP, in crisis settings. A greater quantity and quality of more timely research is needed to ascertain the effectiveness of delivering SRH interventions in a variety of humanitarian crises.
Citation: Singh NS, Smith J, Aryasinghe S, Khosla R, Say L, Blanchet K (2018) Evaluating the effectiveness of sexual and reproductive health services during humanitarian crises: A systematic review. PLoS ONE 13(7): e0199300. https://doi.org/10.1371/journal.pone.0199300
Editor: Ines Keygnaert, Ghent University, BELGIUM
Received: January 3, 2018; Accepted: June 5, 2018; Published: July 6, 2018
Copyright: © 2018 Singh et al. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Data Availability: All relevant data are available in Table 2 included within the paper. Additionally, all included papers in the systematic review are available on the websites of the journals in which they were published.
Funding: This work was funded with support from the Department of Reproductive Health & Research, World Health Organization (to Karl Blanchet). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing interests: The authors have declared that no competing interests exist.
Introduction
The World Health Organisation defines health emergencies as sudden-onset events from naturally occurring or man-made hazards, or gradually deteriorating situations through which the risk to public health steadily increases over time.[ 1 ] It is estimated that 1 billion, or about 14% of the world’s population, live in areas affected by conflict.[ 2 ] According to the United Nations High Commissioner for Refugees, the number of forcibly displaced people has nearly doubled in the past two decades (from 33.9 million in 1997 to 65.6 million in 2016), with numbers remaining at a record high.[ 3 ] Given the changing nature of conflict and protracted crises, the average time spent in displacement has now reached 20 years.[ 3 ] The United Nations Population Fund estimates that of the more than 100 million people in need of humanitarian assistance in 2015, 32 million were women and girls aged 15–49 years.[ 4 ]
Women and girls are disproportionately affected in both sudden and protracted emergencies,[ 5 ] and face multiple sexual and reproductive health (SRH) challenges in conflict and post-conflict settings.[ 6 ] Inadequate or interrupted access to SRH services can also increase the number of people affected, generating a high risk of: mortality or morbidity due to pregnancy-related causes; unintended or unwanted pregnancies due to lack of information or access to contraceptive services; complications related to unsafe abortions; sexual and gender-based violence (SGBV); and an increased incidence of sexually transmitted infections (STIs), including HIV.[ 7 , 8 ] These challenges limit women’s empowerment and their participation in the recovery process, resulting in violations of their human rights, and a reduction in the resources available to alleviate suffering and to be directed towards the process of recovery. Furthermore, there is an economic case for investing in SRH services in humanitarian crises settings, which face financial and other constraints, as it is estimated that for each additional $1 US dollar spent on contraceptive services above the current level of funding in crisis settings, the cost of maternal and newborn health-related care would be reduced by $2.22.[ 4 ]
Over the past two decades, there has been increased attention to SRH in populations in humanitarian settings. In 1994, the importance of refugees’ rights to SRH were articulated and globally agreed in Chapter X of the proceedings of the International Conference on Population and Development in Cairo.[ 9 ] In 1995, the growing awareness of, and commitment to, addressing emergency SRH needs culminated in the formation of the Inter-Agency Working Group (IAWG) on Reproductive Health in Crises, tasked with promoting access to quality SRH care among women and other vulnerable populations impacted by humanitarian crises.[ 10 ] In 1999, the IAWG developed a field manual to provide guidance to field staff on reproductive health interventions in emergencies, and included a chapter on the Minimum Initial Service Package (MISP), to be implemented within 48 hours of the onset of every humanitarian crisis. The MISP aims to facilitate the coordination of SRH services, prevent and manage the consequences of sexual violence, reduce HIV transmission, minimise maternal and neonatal morbidity and mortality, and plan for comprehensive SRH services in the post-crisis phase.
Despite increasing attention to SRH in humanitarian settings, recent evaluations of the MISP have been mixed.[ 11 , 12 ] A systematic review in 2015 [ 13 ] found no peer-reviewed papers that evaluated MISP implementation comprehensively since the first global evaluation in 2004, which identified a low awareness among health actors, and no systematic implementation of the MISP.[ 14 ] The literature is even sparser for studies assessing the effectiveness of SRH interventions including the MISP in humanitarian crises.[ 13 , 15 , 16 ] The only systematic review conducted to date to assess the effectiveness of SRH interventions in humanitarian settings reported a low quantity and quality of evidence, but notably did not comprehensively search for studies focusing on abortion, or aim to disaggregate data by vulnerable sub-populations, e.g. adolescents.[ 15 ]
To build upon progress made and to address evidence gaps, we aimed to consolidate the existing evidence base for the effectiveness of SRH interventions including the MISP from the onset of emergencies by conducting a broader systematic review. Our systematic review aimed to assess the effect of SRH interventions including the MISP and its components reported in the peer-reviewed literature between 1980 and 2017 on a range of health outcomes from the onset of emergencies. As part of this objective, we also aimed to assess issues related to targeting of SRH interventions including the MISP, e.g. whether they are delivered to vulnerable populations such as those with disabilities, sex workers, adolescent girls, and lesbian, gay, transsexual, queer and intersex (LGBTQI) populations.
This systematic literature review adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement ( S1 Appendix ).[ 17 ] The review was registered with the PROSPERO database with identifier number CRD42017082102. The inclusion and exclusion criteria are detailed in Table 1 .
- PPT PowerPoint slide
- PNG larger image
- TIFF original image
https://doi.org/10.1371/journal.pone.0199300.t001
Search strategy and selection criteria
Search terms for SRH were based on the definition of SRH from the International Conference on Population and Development in 1994 [ 9 ] and from the World Health Organisation’s SRH strategies and guidance in 2010 [ 18 ] and 2017 [ 19 ]. SRH refers to a state of physical, emotional, mental and social well-being in relation to sexuality and reproductive health; it is not merely the absence of disease, dysfunction or infirmity. [ 4 , 18 ] International guidelines on SRH in conflict-affected settings include activities related to family planning, abortion, HIV/AIDS and STIs including PMTCT, maternal and newborn health, and SGBV.[ 20 ]
We defined a humanitarian crisis as a serious disruption of the functioning of a community or a society causing widespread human, material, economic or environmental losses that exceed the ability of the affected community or society to cope using its own resources, necessitating a request to the national or international level for external assistance. The crisis situation may either be man-made (e.g. armed conflict) or a natural phenomenon (e.g. drought). Only studies from low-income or middle-income countries were included in this study, as the majority of humanitarian crises occur in these countries, and the resources and strategies available to address them are very different in high-income countries.
A detailed protocol with specific search terms are provided in S2 Appendix , and were generated by the authors and then supplemented by searching for other search strategies used in previous systematic reviews on similar topics.[ 21 , 22 ] We also consulted a trained information science and Cochrane review specialist at the London School of Hygiene and Tropical Medicine to review our literature searching syntax and strategy.
We included studies from peer-reviewed journals across the following four databases: EMBASE, Global Health, MEDLINE and PsychINFO. We complemented searches by screening the reference lists of papers for potentially relevant studies, as well as reference lists of relevant systematic reviews. We also consulted experts on SRH service delivery and research to identify additional research not identified during our systematic search (see Acknowledgments section for list of experts). Selection criteria for literature are detailed in Table 1 .
Data analysis
We downloaded all returned citations from the searched databases into an Endnote library and applied a standard data-screening process ( Fig 1 ). We based the primary and secondary outcomes of interest for inclusion from the IAWG field manual, 14 as it is an established and widely used manual for SRH in crisis-affected settings, and was selected based on discussion with the SRH expert committee. Inclusion and exclusion criteria applied during screening are outlined in Table 1 . Data from the final selected studies were then extracted into a Microsoft Excel database, with data extraction fields including study author and year, setting, crisis type, crisis stage, study design and methods, research setting, health outcomes and intervention descriptions. First round data screening and extraction were independently conducted by NSS, JS and SA. Second round detailed data extraction was conducted independently and in duplicate by NSS and JS.
https://doi.org/10.1371/journal.pone.0199300.g001
We used a narrative synthesis approach due to the heterogeneity of study outcomes, interventions and methods. Findings were synthesised by main SRH outcomes including those included in an earlier systematic review by Warren et al. on SRH interventions in humanitarian settings [ 15 ] i.e. family planning; abortion; prevention, treatment, and care for STIs including HIV/AIDS; maternal, newborn and child health, including obstetric care; SGBV; and studies with cross-cutting SRH themes. These themes were developed iteratively after thematic analysis of the studies’ stated aims and primary reported health outcome of interest.
The quality of reporting in the included studies were assessed using the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) and Consolidated Standards of Reporting Trials (CONSORT) checklists,[ 23 , 24 ] which are commonly used for reporting the quality of observational studies and randomised controlled trials. Both checklists include measures regarding the reporting of participant selection, sample sizes, variables, data sources, bias, descriptive and outcome data, interpretation, and generalisability, among others. In order to further explore the quality of reporting, we awarded papers one point for reporting each of the items on the relevant checklist. When totalled, these points formed the numerator of a proportional score, with the denominator the total number of possible relevant checklist items which varied slightly by study type, i.e. 22 total points for the STROBE checklist, and 25 total points for the CONSORT checklist. NSS, JS and SA conducted quality assessment, who each independently evaluated the quality of all included studies and discussed each discrepancy until consensus was reached. For this systematic review, the study team determined a priori that papers with a score of <33% were considered low reporting quality, moderate quality if 34–66%, and high quality if >67%. These quality thresholds have been used in a previous systematic review on SRH in humanitarian crises settings.[ 22 ]
A total of 14,510 citations were returned from peer-reviewed databases, with one additional study provided by expert recommendation ( Fig 1 ). Following full screening, the review of reference lists, and a review of two existing systematic reviews on similar topics, [ 13 , 15 ] a total of 29 studies met the inclusion criteria ( Table 2 ).
https://doi.org/10.1371/journal.pone.0199300.t002
Of the 29 included studies, 23 (79.3%) were published during the last decade between January 2007 and March 2017. In relation to the geographical distribution of the studies, 17 studies (58.6%) were conducted in Africa, a further nine studies (31.0%) in Asia, one study was conducted in Haiti (3.4%) while the remaining two studies (6.9%) both comprised sites in multiple countries. In relation to the typology of humanitarian crisis, 24 studies (82.8%) were conducted in areas affected by armed conflict, and the two multi-site studies (6.9%) were conducted in areas affected by both armed conflict and natural disasters. The remaining three studies (10.3%) were conducted in areas affected by a natural disaster: the first study focused on the 2005 earthquake in northern Pakistan; the second study focused on the 2013 Typhoon Haiyan in the Philippines; and the third study was conducted in the aftermath of the 2010 earthquake in Haiti.
Of the included studies, over a third (34.5%, n = 10) examined the effectiveness of interventions relating to pregnancy and maternal and newborn health (MNH). Six studies (20.7%) assessed family planning interventions, while a further five studies (17.2%) assessed interventions addressing either HIV or STIs. Three studies (10.3%) examined interventions related to SGBV, while only one study (3.4%) focused on the prevention of mother-to-child transmission of HIV (PMTCT). The remaining four studies (13.8%) assessed a number of cross-cutting SRH domains, of which three studies (10.3%) focused on both family planning and MNH interventions, and one study (3.4%) addressed family planning and abortion care. Notably, no studies addressed interventions aimed at responding to, or the prevention of, vaginal injuries and fistulae.
Study quality
All 29 studies were assessed for quality of reporting using the STROBE checklist. The majority of the observational studies (72.4%, n = 21) were found to be of high quality,[ 25 – 44 ] and the remaining eight studies (27.6%) were of moderate quality.[ 45 – 51 ]
There were common areas in which the included studies in this review provided low quality reporting. All moderate quality quantitative studies stated changes in health outcomes, but statistical associations between the intervention and the outcome were inconsistently reported, i.e. most studies did not report differences between SRH interventions and outcomes. Second, it was also not clear whether relevant confounders and biases were considered during the design of the study and analysis of the data.
Family planning
We identified six studies related to family planning. Along with other SRH outcomes, all six studies assessed contraceptive use in their respective contexts. One paper assessed a refugee-led reproductive health group operating across 48 Guinean refugee camps that recruited refugee nurses and midwives to local health facilities, trained lay women to provide health education and contraception, and to facilitate referrals. Individuals who reported the reproductive health group facilitators as their primary source of information were more likely to be current users of contraception (aOR = 1.3, 95% CI 0.7, 4.2, non-significant).[ 25 ] A home-based counselling and awareness programme for internally displaced women in Sudan led to an increase in the use of modern family planning methods (aOR 2.8, 95% CI 2.0, 4.1).[ 27 ] A programme of mobile outreach and public health strengthening in Uganda led to an increase in the number of women who reported ever using a family planning method (aOR 2.23, 95% CI 1.7, 2.92, P<0.001), and a reduction in the unmet need for family planning from 52.1% to 35.7% (aOR 0.47, 95% CI 0.37, 0.6, P<0.001).[ 26 ] A study in Pakistan providing subsidised healthcare to refugees reported use of contraceptives in the subsidised group (54%) was more than double the use reported in the non-subsidised group (25%), (P<0.001), and reported that the non-subsidised group was more likely to use the oral contraceptive pill (40.7%), whereas the subsidised group was more likely to have tubal ligation (36.7%) (p<0.001).[ 52 ] A multi-country study involving sites in Chad, the Democratic Republic of the Congo, Djibouti, Mali, and Pakistan assessed the impact of a CARE programme that included staff training, supervision in health facilities, the supply of contraceptives, community mobilisation, and awareness raising. An absolute number of 52,616 new users of modern methods of contraception were reported, of which an average of 61% of users across all sites opted for long-acting reversible contraception.[ 46 ] A study in rural Afghanistan assessed the impact of health education and the delivery of injectable contraceptives by community health workers. Over an eight-month period between 2005 and 2006, contraceptive use increased by 24–27% across three sites.[ 45 ]
Pregnancy & maternal and newborn health
We included ten studies reporting on pregnancy or maternal and newborn health-related outcomes. A cross-sectional study amongst internally-displaced women in Darfur, Sudan assessed the impact of a maternal health education programme delivered in the form of home visits. Receiving maternal health education at home was associated with a 43% reduction in the odds of giving birth at home as compared to in a healthcare facility (aOR 0.57, 95% CI: 0.35, 0.93).[ 34 ] Another study amongst internally-displaced women in Darfur found that following the implementation of an interpersonal and mass education campaign, women were more likely to deliver at a healthcare facility (OR 5.4, 95% CI: 4.0, 7.4, P<0.001).[ 35 ] Two studies assessed the effect of referral systems on access to hospital-based care. Following the implementation of an ambulance-based referral system and improved hospital telecommunications in Yirol, South Sudan, the authors reported that 99.1% of the estimated number of women with absolute obstetric indications were treated at the hospital.[ 51 ] A second study examined the impact of a hospital-based vehicle, motorbikes at satellite primary healthcare centres, along with community education activities and facility improvements, on service utilisation and case fatality rates in Bo, Sierra Leone during the period 1992–1993. Service utilisation more than doubled in the period following initiation of the transport system. The case fatality rate declined from 20% to 10% in the post-intervention period. However, there was no significant difference in outcomes between those who were referred using a hospital vehicle and those who used other means to reach the hospital.[ 48 ]
In the Philippines, following Typhoon Haiyan, training of trainers and quality assurance workshops were held, with subsequent improvements in 24-hour access to skilled birth attendants from 84% to 96% at three months (p<0.05).[ 36 ] An evaluation of a programme in West Darfur, Sudan implemented by Medair, including primary healthcare service provision, health promotion activities, and the training of midwives, reported an increase in skilled birth attendance from 35.7% to 52.7% (p = 0.025).[ 37 ] The implementation of a community-based safe motherhood programme in Kabul Province, Afghanistan, delivered by a cadre of newly trained community midwives, reported an increase in the proportion of women receiving antenatal care from 37.3% in 2004 to 91.2% in 2006 (p<0.01), and an increase in the proportion of women delivering at a healthcare facility from 31.3% to 55.2% (p<0.01).[ 33 ]
An evaluation of an International Rescue Committee (IRC) programme, which aimed to reduce maternal mortality among Afghan refugees in Pakistan by establishing emergency obstetric care centres, training community members on safe motherhood, and linking primary care with pregnancy-related education, reported a reduction in the maternal mortality ratio from 291 to 102 per 100000 live births between the first and fifth years of the programme. A reduction in the neonatal mortality rate from 25 to 20.7 per 1000 live births was also reported during the same period, while the proportion of births registered in an emergency obstetric care facility increased from 4.8% in 1996 to 67.2% in 2007.[ 49 ] A study conducted by Save the Children evaluated a birth preparedness package programme implemented in eastern Nepal between 2003 and 2004. The programme involved an educational component delivered by community healthcare workers, and preparation in advance of delivery to optimise maternal and newborn health outcomes. Along with improvements in essential newborn practices, the proportion of women reporting one or more antenatal care visit increased from 60% to 84% (p<0.001), and use of postnatal care within six weeks of delivery increased from 45% to 72% (p<0.001). Changes in the use of a skilled birth attendant were not statistically significant.[ 32 ]
Another study conducted in the Maela refugee camp on the Thai-Myanmar border assessed the effect of a special care baby unit on neonatal and cause-specific mortality rates. Between 2008 and 2011, the neonatal mortality rate declined from 21.8 to 10.7 deaths per 1000 live births (p = 0.03), while cause-specific mortality also fell in relation to the four main causes of neonatal death: prematurity, early onset neonatal sepsis, congenital abnormalities, and jaundice.[ 50 ]
HIV and STIs
A study conducted by Médecins Sans Frontières (MSF) in the conflict-affected region of Bukavu, eastern Democratic Republic of the Congo, reported a median weight gain of 2.5kg and a CD4 gain of 163 cells/ml at 6-months for patients initiated on generic, fixed dose anti-retroviral treatment.[ 47 ] A second study on 24 MSF programmes in 12 countries reported a median 12-month survival of 0.89 (95% CI: 0.88, 0.91) and a median 6-month CD4 gain of 129 cells/mm 3 following the integration of HIV care and treatment programmes with other medical activities.[ 30 ]
A third non-randomised cohort study amongst internally displaced women in Haiti reported an increase in condom use (AOR 4.05, 95% CI: 1.86, 8.83, p<0.001) following implementation of a peer health worker-led intervention involving a video-based session on HIV and STIs, followed by a six-week psycho-educational programme.[ 31 ] A pre-post study of an HIV/AIDS and STI prevention project, including outreach and education activities, in Sierra Leone identified an increase from 38% condom use at last sexual encounter to 68% amongst commercial sex workers, and from 39% to 68% amongst those who identified as members of the military forces.[ 28 ] Following the roll-out of HIV prevention education activities in an urban setting in Sierra Leone amongst adolescents aged 15 to 24 years, contraceptive use increased from 16% to 46% amongst female adolescents, and from 16% to 37% amongst male adolescents (p < .01) [ 29 ].
One study examined the effectiveness of a comprehensive PMTCT programme in conflict-affected northern Uganda. Over the course of a ten-year period, the proportion of HIV-positive women delivering in a health facility increased from 56% to 81% (p = 0.033).[ 44 ]
Sexual and gender-based violence
Three studies, all of which were randomised controlled trials, reported SGBV outcomes. A trial in the Ivory Coast compared the impact of participation in a gender dialogue group and an economic empowerment programme, versus participation only in the economic empowerment programme. Women participating in both the gender dialogue group and the economic empowerment programme were less likely to report economic abuse (OR 0.39, 95% CI 0.25, 0.6, p<0.0001), and were less likely to be accepting of the justification given for violent acts (β = -0.97, 95% CI -1.67, -0.28, p = 0.006).[ 38 ] A second randomised-controlled trial conducted in the Ivory Coast examined the effect of a sixteen-week intimate partner violence prevention programme. Men who received the intervention reported a decreased intention to use physical intimate partner violence (aRR 0.83, 95% CI 0.66, 1.06). Men in the intervention arm reported a greater ability to control their hostility and manage conflict (aRR 1.3, 95% CI 1.06, 1.58). A non-statistically significant difference was identified in reported levels of physical and/or sexual intimate partner violence (aRR 0.52, 95% CI 0.18, 1.51).[ 39 ]
A third randomised controlled trial conducted in the Democratic Republic of the Congo compared the impact of either cognitive processing therapy (comprising one individual session and 11 group sessions), or individual support for female survivors of sexual violence with high levels of post-traumatic stress and combined depression and anxiety symptoms. Both the individual support and group therapy arms reported a statistically significant improvement in all checklist scores (HSCL-25 score, PSTD checklist, and the functional-impairment score) at the end of the intervention, which was sustained at six months post-intervention (p<0.001).[ 40 ]
Interventions targeting outcomes across multiple SRH domains
Four studies reported on outcomes that spanned both family planning and pregnancy and maternal and neonatal health outcomes. A three-tiered community-based intervention in Myanmar employed traditional birth attendants to provide antenatal services and assist with deliveries, while health workers provided antenatal care and family planning supplies, and maternal health workers oversaw the aforementioned activities and attended both normal and complicated deliveries. Use of a modern method of contraception increased from 23.9% to 45.0% (prevalence rate ratio (PRR) 1.88, 95% CI 1.63, 2.17). Unmet family planning needs dropped from 61.7% to 40.5% (PRR 0.65, 95% CI 0.60, 0.72), while birth attendance by someone trained in emergency obstetric care increased from 5.1% to 48.7% (PRR 9.55, 95% CI 7.21, 12.64).[ 42 ] A cross-sectional study reviewed the possible impact of a reproductive health literacy programme amongst refugee women in Guinea. Half of the survey respondents (50%) reported current use of modern contraceptives, while 24% reported using a condom the last time they had sex, of which both findings were interpreted by study authors as an increase since implementation of the reproductive health literacy programme. The study found that 92% of women who reported becoming pregnant since the implementation of the reproductive health literacy programme reported attending at least three antenatal visits.[ 41 ]
In another study evaluating the effectiveness of an emergency obstetric care (EmOC) intervention, a trained physician was posted to a district general hospital in Makeni, Sierra Leone. Additional training was conducted, an unused operating theatre was operationalised, and a generator and blood bank were installed. The proportion of women accessing the hospital increased from 31 in 1990 to 98 in 1995, with a reduction in the case fatality rate from 32% to 5%. Notably, 444 induced abortion-related procedures were performed, compared with only 22 in 1990.[ 53 ]
Following the 2005 earthquake in Pakistan, a four-year project was initiated by USAID with the aim of improving the management capacity of district health authorities, improving access to primary healthcare services, increasing community participation in service management, and improving household-level knowledge and health-seeking behaviour. The study found non-statistically significant increases in the use of modern contraceptives (18% to 22%), at least one antenatal care visit (70.3% to 73.6%), and presence of a skilled birth attendant (36% to 38%). The study also found a statistically significant increase from 25% to 33.3% (p<0.01) in women receiving postnatal care.[ 43 ]
This review identified 29 studies assessing the effectiveness of SRH interventions in humanitarian crises published between 1980 and 2017. We found high quality evidence for specific interventions appearing to have improved SRH outcomes, including home visits and peer-led educational and counselling, training of lower-level health care providers, the use of CHWs to promote SRH services, a three-tiered network of health workers providing SRH services, the integration of HIV and SRH services, and men’s discussion groups to address intimate partner violence. We found moderate quality evidence to support transport-based referral systems, community-based SRH education, CHW delivery of injectable contraceptives, wider literacy programmes, and birth preparedness interventions. We identified no studies focusing on adolescents, LGBTQ populations or people with disabilities. Additionally, no studies measured the effectiveness of interventions addressing the comprehensive clinical management of rape, or the response to, or prevention of, vaginal injuries and fistulae.
The quality of studies included in the review was variable. Three quarter of the studies (72.4%, n = 21) used either experimental designs, i.e. randomised controlled trials, or quasi-experimental study designs, which provided some statistical measure of difference between intervention and outcome. However, where appropriate, there was often insufficient adjustment for potential confounders Evidence of attribution was particularly weak, with the vast majority of studies using cross-sectional and pre-post study designs with no control.
Although the majority of studies (72.4%, n = 21) were graded as high quality, the application of the STROBE checklist revealed a number of commonly recurring methodological weaknesses. Blinding was rarely used, including in the randomised controlled trials, which risks reporting bias of health outcomes. There was a lack of controlled studies, limited appreciation of clear exposures and confounders, absence of reporting on sampling methods, and an inadequate handling of bias. However, it is recognised that for a number of SRH interventions and in certain humanitarian contexts, blinding or use of control groups is not possible or appropriate. There was also limited use of stratification, for example by gender or age, and so potentially differing health outcomes in more vulnerable groups is missing.
We recognise that there are many logistical and ethical challenges related to SRH research in humanitarian crises. However, the more developed evidence base for other health outcomes in similar crises settings, e.g. mental health and communicable diseases,[ 16 ] signals that conducting more rigorous research in humanitarian contexts is possible. Given the lack of longitudinal data or studies with an adequate control comparison group, innovative ways of collecting data, e.g. using information and communication technologies (ICT) widely used by many conflict-affected populations such as WhatsApp, should be tested. These data collection methods may prove beneficial for researchers, healthcare providers, and organisations seeking to collect health outcome data at the individual-level, and from populations on the move who have traditionally been challenging to follow up. There is also a need for innovation in establishing stronger referral and follow-up systems in crisis settings to ensure health outcomes used to assess effectiveness are as close to its true effectiveness as possible. Researchers should also consider use of alternative study designs where standard RCTs are not operationally or ethically possible. Adjusted approaches such as stepped wedge trial designs could be used more widely in order to establish a counterfactual through the use of a control group, while remaining operationally and ethically acceptable. Additionally, no studies reported measuring outcomes beyond the study period, signalling the need to have data on longer-term effects of SRH interventions on target populations affected by humanitarian crises. Despite a well-established SRH evidence base in stable settings, further robust research is needed to determine effective and cost-effective interventions to improve SRH outcomes in populations in crisis settings.
This systematic review had a number of limitations. Caution must be exercised in generalising our study’s findings, as the specific contexts, and enabling and restrictive factors to intervention delivery and effectiveness, varied widely between studies. Although we based search terms and developed inclusion criteria to best address our research aim, our search may have missed identifying additional papers from relevant SRH domains. We also did not conduct a search for grey literature. Our strict study design criteria also excluded qualitative studies, which form a key component of the literature in this field. However, we only included quantitative studies demonstrating quantitative change over time with the aim of extracting a certain type of evidence to inform our findings on the effectiveness of SRH interventions in crisis settings. Additionally, only papers written in English and French were included. We applied a narrative synthesis approach to the findings, as we were unable to conduct a meta-analysis due to the heterogeneous nature of intervention types, indicators and methods used in the included studies. We used the STROBE checklist to assess the quality of reporting in studies, but a more specialised quality assessment tool such as the Newcastle Ottawa Scale or Cochrane Collaboration's tool for assessing risk of bias would have provided a more robust review of quality.[ 54 ]
This systematic review found high quality evidence to support improved SRH outcomes through home visits and peer-led educational and counselling, training of lower-level health care providers, CHWs to promote SRH services, a three-tiered network of health workers providing SRH services, the integration of HIV and SRH services, and men’s discussion groups for the reduction of intimate partner violence. However, the types of study design including many studies with no control group, the limited use of statistical data, and the variable quality of studies, signal that caution must be exercised in the interpretation of these results. There is a need for a higher quantity and quality of more timely research on the effectiveness of SRH interventions in humanitarian crises, in particular focusing on outcomes amongst adolescents, and studies that focus on safe abortion care, post-abortion care, vaginal injury and fistulae, the prevention of SGBV, and the comprehensive clinical management of rape.
Supporting information
S1 appendix. prisma checklist..
https://doi.org/10.1371/journal.pone.0199300.s001
S2 Appendix. Systematic review protocol.
https://doi.org/10.1371/journal.pone.0199300.s002
Acknowledgments
We thank Maria Rodriguez, Loulou Kobeissi, Adam Poulter, Robyn Drysdale, Rajrattan Rokhande and Sara Nam for reviewing the study protocol. The authors alone are responsible for the views expressed in this manuscript and they do not represent the views, decisions or policies of the institutions with which they are affiliated.
- 1. World Health Organization. Emergency Response Framework 2014 [5 December 2017]. Available from: http://apps.who.int/iris/bitstream/10665/89529/1/9789241504973_eng.pdf?ua=1 .
- 2. State of world population New York: United Nations Population Fund; 2015 [14 November]. Available from: http://www.unfpa.org/sites/default/files/sowp/downloads/State_of_World_Population_2015_EN.pdf .
- View Article
- PubMed/NCBI
- Google Scholar
- 4. Adding It Up: The Costs and Benefits of Investing in Sexual and Reproductive Health: Guttmacher Institute; 2017 [14 November]. Available from: https://www.guttmacher.org/fact-sheet/adding-it-up-contraception-mnh-2017 .
- 6. United Nations. Report of the Secretary-General on women and peace and security New York: United Nations; 2014 [21 November 2018]. Available from: http://www.un.org/ga/search/view_doc.asp?symbol=S/2014/693 .
- 9. Report of the International Conference on Population and Development. International Conference on Population and Development; New York: United Nations 1994.
- 14. United Nations High Commissioner for Refugees. Reproductive Health Services for Refugees and Internally Displaced Persons: Report of an Inter-agency Global Evaluation. 2004.
- 18. World Health O. Measuring sexual health: Conceptual and practical considerations and related indicators. 2010.
- 19. World Health O. Sexual health and its linkages to reproductive health: an operational approach. Sexual health and its linkages to reproductive health: an operational approach2017.
- Open access
- Published: 09 February 2021
A qualitative exploration of contraceptive use and discontinuation among women with an unmet need for modern contraception in Kenya
- Susan Ontiri ORCID: orcid.org/0000-0001-7622-5714 1 , 2 ,
- Lilian Mutea 3 ,
- Violet Naanyu 4 ,
- Mark Kabue 5 ,
- Regien Biesma 2 &
- Jelle Stekelenburg 2 , 6
Reproductive Health volume 18 , Article number: 33 ( 2021 ) Cite this article
11k Accesses
19 Citations
2 Altmetric
Metrics details
Addressing the unmet need for modern contraception underpins the goal of all family planning and contraception programs. Contraceptive discontinuation among those in need of a method hinders the attainment of the fertility desires of women, which may result in unintended pregnancies. This paper presents experiences of contraceptive use, reasons for discontinuation, and future intentions to use modern contraceptives.
Qualitative data were collected in two rural counties in Kenya in 2019 from women with unmet need for contraception who were former modern contraceptive users. Additional data was collected from male partners of some of the women interviewed. In-depth interviews and focus group discussions explored previous experience with contraceptive use, reasons for discontinuation, and future intentionality to use. Following data collection, digitally recorded data were transcribed verbatim, translated, and coded using thematic analysis through an inductive approach.
Use of modern contraception to prevent pregnancy and plan for family size was a strong motivator for uptake of contraceptives. The contraceptive methods used were mainly sourced from public health facilities though adolescents got them from the private sector. Reasons for discontinued use included side effects, method failure, peer influence, gender-based violence due to covert use of contraceptives, and failure within the health system. Five reasons were provided for those not willing to use in the future: fear of side effects, cost of contraceptive services, family conflicts over the use of modern contraceptives, reduced need, and a shift to traditional methods.
This study expands the literature by examining reasons for contraceptive discontinuation and future intentionality to use among women in need of contraception. The results underscore the need for family planning interventions that incorporate quality of care in service provision to address contraceptive discontinuation. Engaging men and other social influencers in family planning programs and services will help garner support for contraception, rather than focusing exclusively on women. The results of this study can inform implementation of family planning programs in Kenya and beyond to ensure they address the concerns of former modern contraception users.
Peer Review reports
Use of contraceptive methods allows spacing of pregnancies or limiting family size, enabling individuals and couples to fulfill their fertility desire by choosing if and when to become pregnant. Contraceptive use not only has positive effects on health-related outcomes, such as improved maternal and child health [ 1 ] but also improves schooling and economic outcomes for girls and women [ 2 ]. Global trends have shown an increase in contraceptive uptake, however, many women, approximately one out of three, discontinue their method within a year [ 3 , 4 ]. Contraceptive discontinuation is an important determinant of contraceptive prevalence, as well as unintended pregnancies, and other demographic impacts as it increases the unmet need for family planning (FP). Several studies have found that contraceptive abandonment and failure contribute substantially to the total fertility rate, unwanted pregnancies, and induced abortions [ 3 , 4 , 5 ]. Analysis of data from 36 developing countries revealed that over one-third of unintended pregnancies resulted from women who had discontinued the use of contraception [ 5 ]. Unintended pregnancies have negative consequences on the health and well-being of women and their families as they can lead to maternal morbidities and even death. Besides, it is documented that children born from unintended pregnancies are: less likely to be breastfed, more likely to be stunted, at risk of a lack of parental love, and at higher risk of child mortality than children from wanted pregnancies [ 6 ].
An analysis of Demographic and Health Surveys conducted by Curtis et al. demonstrated that women’s socio-demographic characteristics—age, education, place of residence, and economic status—are the determinants associated with contraceptive discontinuation [ 7 ]. Even though studies indicate that women with higher levels of education and those residing in urban residences are more likely to discontinue their initial method, additional analyses reveal that these women are more likely to switch than stop after discontinuing a method [ 7 , 8 , 9 ]. This could be because they are enlightened on their contraceptive choices and will discontinue and switch if a particular method does not suit them since they can also easily access the contraceptive services due to shorter distances to health facilities.
Researchers continue to investigate why a woman or a couple would discontinue the use of modern contraception while still in need. Past studies show side effects and health concerns have been the main causes of contraceptive discontinuation [ 3 , 4 , 10 ]. Indeed, side effects account for more than half of the reasons for discontinuing contraceptives while still in need [ 9 , 11 ].
Kenya has implemented a strong national family planning (FP) program since it was launched in 1967 [ 12 ]. Over the past five decades, the country has developed FP/reproductive health policies, strategies, and guidelines and implemented programs aimed at increasing access and utilization of modern contraceptive methods among women of reproductive age and supporting men's involvement. These efforts have borne fruit; the current data estimates a contraceptive prevalence rate of 62.8%, which is mostly driven by the use of modern methods at 60.7% [ 13 ]. However, more than one-third of all pregnancies in Kenya are unintended and one in three women discontinue use of contraceptives by 12 months [ 14 ]. Like other countries, the main reason cited in Kenya for discontinuation is side effects, predominantly side effects associated with hormonal contraception [ 14 ]. Studies have linked poor quality of care, particularly inadequate counseling on side effects with contraceptive discontinuation [ 4 , 15 ]. For instance, data from round 5 to round 7 of Kenya’s Performance Monitoring and Accountability 2020 surveys indicate a glaring gap in the quality of FP services provided in health facilities. Only two-thirds of women were informed about side effects by service providers, with slightly more than half being informed about what to do in case of side effects [ 13 , 16 , 17 ].
Whereas the predictors of contraceptive counseling have been established by several quantitative studies [ 3 , 4 , 18 ], there is a paucity of information to understand the lived-in experiences of women who discontinue the use of contraceptives while still in need. This paper reports qualitative results from in-depth interviews and focus group discussions with discontinuers. The interviews and discussions explored experiences with previous use of modern contraceptives, reasons for discontinuation, and future intention to use contraceptives among discontinuers.
Study design and setting
A cross-sectional qualitative study was conducted as part of a formative assessment in a 24-month longitudinal study on evaluating the dynamics of contraceptive use, discontinuation, and switching in Kenya. The longitudinal study is being conducted in Kitui and Migori, rural counties in Kenya. The two counties have a diverse method mix; Migori’s mCPR is mostly driven by long-acting reversible contraceptives, at 72% while in Kitui, short-term methods are more popular, at 64% [ 14 ]. Details of the longitudinal study, including the study setting, have been published elsewhere [ 19 ]. Ten public health facilities, five in each county were purposively selected based on high FP caseload. The 10 facilities were located in 10 different sub-counties. Routine service statistics revealed that these facilities provided the highest number of contraceptive services in their respective sub-counties. Out of the ten facilities, 2 were county hospitals, 5 sub-county hospitals, 2 health centers, and 1 dispensary. The consolidated criteria for reporting qualitative research (COREQ) was used in this paper [ 20 ]. The completed checklist is available in Additional file 1 .
Study participants
Since the main objective of this study was to explore the experience with contraceptive use and discontinuation among discontinuers, participants who met the following inclusion criteria were selected: women of reproductive age between 15 and 49 years of age, who were sexually active, did not desire pregnancy, and had been but were currently not using modern contraception. The men who were interviewed to explore their perspective on contraceptive discontinuation were purposively selected since they were spouses of the women who met the inclusion criteria. Data collection included FGDs with adolescent mothers aged 15–19 years and women over 20 years and IDIs with couples and adolescent girls. Recruitment of study participants stopped once data saturation was achieved, that is when no new information was derived from the interviews and focus group discussions. In total, 42 data collection sessions (12 FGDs and 30 IDIs) were conducted with 135 study participants-105 in FGDs and 30 in IDIs. (Table 1 ).
Recruitment strategy
The study team selected community health volunteers (CHVs) who were providing health information including family planning to households within the catchment area of the study facilities. The CHVs were trained on the inclusion criteria and thereafter, mobilized and screened community members within their catchment area before referring them to the study staff who contacted, further screened, and recruited those eligible into the study. For couples, the CHV would approach the woman first to establish eligibility, before contacting the spouse. Both partners had to agree to participate before inclusion in the study.
Data collection
Data collection was conducted from May to July 2019. The data collection team was comprised of 10 research assistants, (seven females and three males) who had undergraduate training in Anthropology or Sociology. The team was selected based on their experience conducting qualitative studies. They further received an additional 5-day refresher training before data collection. They worked under the supervision of the lead author. Respondents were not known to the interviewers before the data collection sessions. Written consent was obtained from the participants to conduct and audio-record the data collection sessions. The time and place of the interviews were determined based on the convenience of the participants. The venue for the FGD data collection sessions was community halls while the IDIs were conducted at the participants’ homes. All participants were aware that the study was being conducted to explore their perspective and experience with contraceptive use and discontinuation as part of a formative assessment to improve the quality of family planning services provided.
Semi-structured topic guides covering FP topics for the various audiences were developed and piloted before use. The FGD guide included open-ended prompts related to knowledge and perception of contraceptives, use of FP with their community, and reasons for contraceptive discontinuation, including influencers. The study had IDI guides for the adolescent girls (15–19 years) and for married couples (18–49 years), husbands and wives were interviewed separately. The former group was asked about their knowledge and perceptions around sexual and reproductive health and contraceptive use, experience using contraceptives, and contraceptive discontinuation. The married couples shared their knowledge, perception, and decision-making experiences using contraceptives; FP use and discontinuation; and couple involvement in contraceptive use and discontinuation. The file showing the topic guides used in this study is provided in Additional file 2 .
Two trained interviewers were present at each FGD—one as a session moderator and the other as a note-taker. For the IDIs, only one trained moderator was present for the conversation. No observer was present during data collection. The FGDs and interviews were conducted in local dialect (Kamba and Dholuo) and Swahili. All the interviews were audio-recorded, and field notes were taken for each focus group session. The interview sessions lasted between 30 and 90 min. The data collection team debriefed after the end of each session. Interim findings were discussed weekly by the team and interview guides were modified and revised as needed. At the end of data collection, no new themes were emerging and data saturation had been achieved.
Data analysis
The digital recordings of IDIs and FGDs were transcribed verbatim, translated into English, and analyzed using NVivo 11. Data were analyzed thematically following the approach of Braun and Clarke to identify, analyze, and report patterns within the data [ 21 ]. Coding and theme development were directed by the content of the data (inductively) [ 21 ]. A final agreed thematic framework was applied to all interviews. Transcripts were not returned to participants in advance of coding. Data analyses were performed by two researchers (VN and SO) with in-depth knowledge of qualitative analysis who were supported by two analysts to ensure timely coding and validation of the coding frame. The team identified themes from reading and rereading the transcripts, noting any similarities and differences between and within participants’ accounts. The preliminary findings were shared with some of the study participants for validation.
Ethical considerations
This study was guided by a protocol that was approved by the Kenya Medical Research Institute Institutional Review Board and the Johns Hopkins Bloomberg School of Public Health Institutional Review Board. Participants gave informed written consent/assent to participate in the study. Protection and confidentiality of participants was ensured through conducting data collection sessions in private settings, maintaining confidentiality, and limiting access to study information to only authorized personnel.
The demographic characteristics of the 135 study participants are shown in Table 2 . The majority of the participants were adolescents and youth aged 15–24 years at 51%, had primary education 53%, were farmers 32%, and had one to two children (Table 2 ). The findings from the two study sites were comparable, with no major differences.
Study findings are provided in four themes below: (1) motivation for modern contraceptive use; (2) sources and decision-making for previous contraceptive used; (3) barriers to sustained use of contraceptives; and (4) future intention to use contraceptives.
Motivation for modern contraceptive use
The study explored the participant’s motivation for use of a contraceptive prior to discontinuation. Generally, there was strong consensus among all the study participants that the reasons for using contraceptives were to plan for the number of children they wished to have, and prevent pregnancy. Adolescent participants further noted that the greatest motivation for using contraceptives was to prevent pregnancy so as to pursue studies; they wanted to avoid unplanned pregnancies that might result in having to drop out of school and take on parental responsibilities they had not envisioned.
Economic reasons appeared to be the major impetus for use of contraceptives by adolescent mothers, older women, and married couples, as most participants shared similar sentiments on the need to have children they can manage to raise as illustrated by the following quote:
“We are able to space out the children and able to provide the right foods to the children so that they can be healthy because our incomes are low.” (FGD, Female).
Many participants reported that their motivation for use of contraceptives was to space their pregnancies to allow the healthy growth of children so they could get enough attention, nutrition, and care from their parents. A few married women noted, where couples were experiencing marital conflict, women used contraceptives to avoid getting additional children that they would need to support on their own.
Sources and decision-making for previous contraceptive used
The majority of participants interviewed indicated that they got their contraceptive method from public health facilities. Some, especially adolescents, got their contraceptive methods from private facilities, specifically chemists or pharmacists. Most older respondents indicated that they had opted for injectables and implants, while use of pills was mainly mentioned by adolescents.
“I bought my pills from the pharmacy shop in town” (IDI, Adolescent, Female).
The study findings revealed that before using contraception, most women sought the opinions of partners, peers, or family friends. For adolescent mothers, their mothers were mentioned as helpful in decision-making and accessing contraceptives. Most partners were involved in decision-making about uptake of FP before initiation of a method, while some were engaged after the FP method was started. However, some female participants stated that they had used contraception covertly due to non-supportive spouses or relatives, particularly the in-laws who threatened to report them to their partners.
Barriers to sustained use of contraception
The study further explored the reasons why women did not continue using a contraceptive method yet they still had a need for contraception. Reasons for discontinued use of contraceptives were manifold; five main sub-themes emerged: side effects, method efficacy, peer influence, gender-based violence, and health system factors.
Side effects of contraceptives
Across all the study groups, side effects resulting from use of contraception were repeatedly mentioned among the reasons for discontinuation. The leading side effect was irregular bleeding patterns presenting as menorrhagia (heavy menstrual bleeding) or amenorrhea (absence of menstrual bleeding). This was mainly experienced from the use of hormonal methods, and in particular injectables and implants. For example:
“When I used the three-months injection, I was bleeding excessively. Sometimes I would feel dizzy while walking. The bleeding would even continue for a month without stopping. So, I decided to stop using it.” (IDI, Female).
Heavy bleeding was cited to interfere with the participants’ social and economic lifestyle. The majority of the female participants who reported increased bleeding indicated that they were unable to carry out their economic activities since they were weak as a result of the increased menstrual flow. Another recurrent consequence of the increased bleeding was the interference with their sexual life:
“The reason I chose to stop using depo is for one reason. Sometimes my husband may have the desire to get intimate with you but you cannot, because of the bleeding. Whenever I want us to get intimate he declines because it is so much blood that is why he told me to try quitting it.” (IDI, Female).
On the contrary, some respondents reported that the absence of menstrual bleeding was what triggered discontinuation since they did not know whether they were still fertile or were pregnant.
“When I started using implants, my periods did not come for eight months, then it came back only for two days and disappeared again. I decided to stop using a contraceptive since I was always wondering whether I was pregnant.” (FGD, Adolescent).
Other side effects that led to discontinuation, albeit less frequently mentioned across the various study groups, included weight changes, dizziness, and low sexual libido.
“ My friend who was using the one for three years told me she stopped because she didn’t have an appetite for having sex, so it was raising issues between her and her husband.” (FGD, Adolescent).
Some study participants observed that experiences from other women influenced contraceptive use or discontinuation. Several FGD participants indicated that women discontinued the use of contraceptive methods after learning about side effects experienced by their friends. This prompted even those who were not experiencing the same to discontinue out of fear.
Contraceptive method efficacy
Contraceptive efficacy was a concern mentioned mostly by married couples. Respondents reported method failure whereby women got pregnant unexpectedly while still on a contraceptive method:
“One year after using an implant, I started becoming sick. When I went back to the hospital, I was tested and the results came out that I was four months pregnant, and at the same time I still had the implant in my arm.” (FGD, Female).
“I have a friend; she was using the one for 3 months. After sometime, she was shocked that she was pregnant. So, she decided that she will not use it because even if you use it you still get pregnant.” (FGD, Adolescent).
Several participants revealed that they decided to discontinue use of contraceptives after learning about cases of method failure among women who were using similar methods. On several instances, inconsistent use of contraceptive, especially short-term methods, that resulted in pregnancies were reported as method failure by some participants:
“The one for three months confused her a lot, it came to end without her knowing and she forgot to go back to the clinic for another injection. She became pregnant and then it surprised her. We had tried using it for a long time and I told her that she was using a method of a shorter duration and when it ended she became pregnant without planning.” (IDI, Male).
Covert use of contraception resulting in gender-based violence
Covert use of contraception was common due to lack of spousal support for use of a modern method. Across all the study groups, the participants shared their experiences or cases of other women who discontinued contraceptive use because their partners learned that they were using it covertly. Cases of gender-based violence directed at women by their partner after learning their use of modern contraceptive methods, further solidified their resolve to discontinue as illustrated by this experience:
“Another woman in our village went and got an implant without her husband’s knowledge. When the husband learned of this, he took a knife and removed it from her arm. This made my friends and me afraid, so we decided to just remove it for fear of what our husbands would do if they find out.” (FGD, Female).
Health system factors as a barrier to continuation
Health care system factors were repeatedly mentioned as reasons for discontinuation. Stock-outs of preferred methods during contraceptive initiation or resupply prompted women to either take alternative methods or leave without one. Provider bias that resulted in women taking up methods that they did not approve of came up as a sub-theme particularly by younger women, as shown in the quote below:
“I told him [the provider] I wanted depo and he said that the government does not advise the use of injection, and he refused to put it on me. He convinced me to take up an implant, which I did, but I went to another facility to have it removed.” (FGD, Female).
There were mixed experiences regarding FP counseling, particularly on side effects. Several respondents noted that they got adequate counseling by the health care providers during the initiation of a method; however, some mentioned that they were not informed of any potential side effects that could result from use of contraception.
“When I started using them, the doctor explained to me about the advantages and disadvantages of the various methods of family planning, such that, I know the goodness and effects of the method I am using.” (FGD, Female).
Future intentionality to use contraception
The study explored whether the respondents would consider using modern contraceptives again. Several respondents indicated willingness to use at some time, but some were hesitant. Those who would consider using an FP method again said they would consult widely, select a method with fewer side effects, and one with a longer duration. For those who were doubtful and not considering using FP, five reasons were provided.
First, there were fears about negative side effects. Women indicated that the fear of experiencing another side effect after discontinuation led them to decide not to take up any other modern method despite the counseling that they got from health care workers who were advising them on method switching. One woman shared her experience:
“These medicines bring problems. I stayed with the one injection for a while and every time I would feel sickly, weak, back pains at all times, bleeding from Monday to Monday. I came to the hospital and asked them to remove it. They asked me what the problem was, that they will give me another one, but I did not want one. So that is why I stopped using.” (FGD, Female).
Second, cost was cited as a barrier for continued use. Respondents indicated that the direct and indirect costs associated with uptake of contraceptive services hindered their intention to use. The cost barrier was mainly mentioned for short-term methods that require frequent resupply at facilities, hence, women had to make multiple visits to the facility. Several concerns were also raised regarding the removal of intrauterine contraceptive devices or implants after experiencing side effects. An important issue that participants highlighted was the cost incurred for the removal of a method, which caused women to fear the selection of another method in case they experienced side effects with that method.
“If you go to the facility before the expiry date, you are asked to pay 200 shillings, regardless of the side effects experienced. I wonder why they charge for removal yet they gave it for free. After that one fears to take up another method.” (FGD, Female).
Lastly, FP use caused conflicts in families. Women indicated lack of support from their partners and relatives impeded their intention to use contraception. It was evident that even though the women felt a need to space or limit their family size, that decision was mainly made by their partners. Other women, who had previously used the method covertly and had been discovered by their spouses or relatives, mentioned they could not use the method for fear of gender-based violence. This quote buttresses the point:
“My husband threatened to beat me also if he ever found me using a method. This was after he had observed a disagreement between our neighbors (couple), over the discreet use of contraceptives that ended up with the lady being hit by her husband. I decided to stop using to avoid such an occurrence. ” (IDI, Female).
This qualitative study aimed to explore the dynamics of contraceptive use and discontinuation among women with unmet need for contraceptives in the rural counties of Migori and Kitui, Kenya. A large and diverse group of adolescents, women, and couples who reported contraceptive discontinuation while still in need of a method provided insights on their experiences, perspectives with contraceptive use and reasons for discontinuation. Direct quotes of study participants about their experiences with FP use that culminated in discontinuation have been presented to deepen understanding of participants’ experiences [ 22 ]. From the study findings, it is evident that all the respondents chose to use contraceptives with the conviction that by using a modern method, they would be able to prevent pregnancy or plan when to have children, determine how far apart they want their children to be, and when to stop having children. However, this desire was not fully realized as they discontinued use of the contraceptives while still in need, which added to the pool of women of reproductive age with unmet need for FP.
There were numerous challenges faced by women using contraceptives that prompted them to discontinue their use. As noted in prior studies, side effects play a major role in reported decisions to discontinue [ 4 , 23 , 24 ]. Our study revealed that the most common side effect leading to contraceptive discontinuation were changes in users’ bleeding patterns, findings which are consistent with studies conducted across different parts of the world [ 18 , 25 , 26 ]. Irregularity of bleeding negatively impacts the well-being of women, mainly due to the social consequences, which could explain the low tolerance with contraception when such side effects are encountered. Studies have revealed that women, especially in the sub-Saharan region, believe that menstrual bleeding is a sign of fertility, hence any change that leads to reduced or no bleeding is frowned upon [ 27 , 28 ]. Conversely, increased bleeding impacts women’s socio-economic activities and sexual relationship with their partners [ 28 , 29 ].
Our findings thus provide strong support for addressing side effects experienced by women through management when they occur or being provided options for method switching to ensure the women continue to harness the full benefits of contraception. This can be achieved by conducting client follow-up by service providers to periodically assess the level of satisfaction with the contraceptive method while addressing issues that might prompt clients to discontinue. Proper counseling of clients, and their partners, is crucial to promote continuation with use of modern contraceptive methods as the users are made aware of the contraceptive’s mechanism of action, possible side effects, and what to do when they experience side effects. Helping women understand typical bleeding changes associated with their contraceptive methods could lead to greater acceptance of the changes, increased method uptake, improved satisfaction, and higher continuation rates [ 30 ]. Therefore, capacity building of health care providers on contraceptives should not just focus on the technical skills on insertion and removal (particularly for long-term methods), but also on contraceptives’ mechanisms, how they work, to ensure that providers are well versed on the potential side effects for each method. This is supported by evidence from studies in Madagascar and Ghana that revealed providers were not well informed on the physiological effects of contraception and how to manage side effects [ 4 ]. This resulted in inadequate counseling of women experiencing the side effects; women were counseled to switch to another method instead of being reassured that side effects would settle down over time or being offered medication to control some side effects [ 4 ]. This could be attributed to inadequate training content on side effects. A recent review of FP counseling, training, and reference materials revealed that bleeding changes are insufficiently addressed in capacity building resources and counseling tools for health care providers [ 29 ]. This is alarming, considering that the leading reason for discontinuation has been changes in bleeding pattern. Skilled counseling for side effects, particularly bleeding irregularities, can only be achieved if training materials for health care providers incorporate this information, information that will improve the quality of counseling by health care providers.
Contraceptive method failure was one of the reasons for discontinuation in this study. Method failure is a factor of either failure of a method to work as expected or incorrect/inconsistent use of a method by the user. In low- and middle-income countries, 74 million unintended pregnancies occur annually, of which a sizable share, 30%, are due to contraceptive failure among women using some type of contraceptive method [ 31 ]. Each contraceptive method has a Pearl Index number that reflects pregnancy rates during perfect and typical use, with use of long-term method conferring higher efficacy than short-term methods [ 32 ]. Whereas all contraceptive methods have some degree of failure, even during perfect use, failure rates can be reduced when individuals are sensitized on the proper use of contraception to ensure the method is used correctly and consistently. Provision of clear information about the risks and benefits of all available methods is crucial in facilitating informed contraceptive choice so women can make an educated choice for their preferred methods, which may reduce discontinuation.
Other reasons for contraceptive discontinuation, such as lack of support from partners and other social networks, are also corroborated in researches previously conducted in Kenya [ 28 , 33 ]. In our study, the decision to use or not use contraceptives was still primarily made by men. Although women made solo decisions on FP, they were heavily influenced by their spouses’ preference and would stop using if they thought it would bring marital conflicts. Opposition to contraceptive use by husbands appears to stem from the fear of side effects and the perception that women who use FP are more likely to be promiscuous. Additionally, Kenya being a highly patriarchal society, decision-making around the desired number of children mainly lies with the male partner. FP programs have mainly targeted women with information to promote uptake since they are the ones who face the risk of pregnancy and childbirth. Unfortunately, these programs have left out men, who are in most instances, the decision-makers in male-dominated societies, like most countries in the sub-Saharan region [ 34 ]. The findings from this study reveal the power dynamics when it comes to a couple’s decision to use contraception. This underscores the need to meaningfully involve men in FP programs by informing them of the health, economic, and social benefits realized from proper and consistent use of contraception so they can optimize use of FP services. Demand generation strategies that employ the use of positive deviants, satisfied users, and other key influencers, such as mothers-in-law, may lead to an increase in contraceptive uptake and enhance continuation.
This study indicates that the costs associated with consistent use of FP methods hinder their continued use. Promoting uptake of LARC methods will address the cost associated with the use of short-term method—LARCs have been shown to be more cost-effective and do not require frequent visits to facilities [ 35 ].
Our study also revealed punitive measures women faced, especially those on LARCs, when they wanted to switch to another method before its expiration. Allowing for method switching is indicative of strong FP programs that have an adequate range of methods and a flexible environment to meet women’s needs. Due to the health and social concerns that contraceptive use may confer on individuals, women may try different methods before settling for their preferred option. The health system should have a supportive policy environment that accommodates such needs of women by: instituting guidelines that prohibit penalization for method switching; addressing commodity stock-outs and ensuring sufficient method mix through increased financing of FP programs; and sensitizing providers on the importance of method switching by women who are not satisfied with their methods. Additional studies are needed to document the implications of frequent method switching on commodity security in countries that continue to face widespread stock-outs of contraceptive methods.
The study’s main strength was documenting the experiences of contraceptive use and discontinuation among discontinuers themselves. However, qualitative studies have limitations related to validity, subjectivity, and reliability. To address these issues, efforts were made to increase the rigor and trustworthiness of the findings through the selection of participants with a range of backgrounds and experiences with the guidance and supervision of experts, as well as external review. Information was not collected on the number of eligible participants who refused to participate in the study. Despite this, our study benefits from including a large number of participants, diverse in terms of age, gender, ethnicity, and location, and utilizing different data collection methodologies (FGDs and IDIs) to enrich the findings.
Conclusions
Our study, conducted in two rural counties in Kenya, revealed a number of important findings regarding factors influencing contraceptive use and discontinuation. The participants in this study had a common motivation for using contraception, to avoid pregnancies, however, side effects were a major hindrance in continued use of contraception. Covert use of contraception resulted in discontinuation when it was discovered and, in some instances, led to gender-based violence. Decision-making on contraception, method to use, and the number of children to have, was jointly done by couples or made by the husband. Reasons for discontinuation, specifically on side effects, were influenced by the husbands.
As contraceptive use in a population increases, success in avoiding unintended pregnancies depends less on initial contraceptive uptake and more on effective and persistent use. Enhanced efforts are needed to design and implement programs that focus on contraceptive discontinuation among women with unmet need for FP. Health care providers offering FP services should be well versed with the mechanism of action for the various contraceptive methods, and incorporate quality of care in the provision of contraceptive services. Additionally, contraception technological advancement is urgently needed to expand the method mix and to develop methods that have fewer side effects and side effects that can be more easily tolerated. This will go a long way in promoting continuation of contraceptive use, as indicated by a majority of our study participants who were willing to consider future use of contraception methods with fewer side effects. Findings from this study, as well as other studies, confirm the importance of engaging men and other social influencers in FP programs by educating them on the socio-economic and health benefits of family planning and dispelling any myths and misconceptions to create a social environment that supports use of modern contraception.
Availability of data and materials
The data used and analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
Consolidated criteria for reporting qualitative studies
Community health volunteers
Focus group discussions
Family planning
In-depth interviews
Long-acting and reversible contraceptive
Total fertility rate
Starbird E, Norton M, Marcus R. Investing in family planning: key to achieving the sustainable development goals. Glob Health Sci Pract. 2016;4(2):191–210.
Article Google Scholar
Canning D, Schultz TP. The economic consequences of reproductive health and family planning. Lancet. 2012;380(9837):165–71.
Ali MM, Cleland JG, Shah IH. Causes and consequences of contraceptive discontinuation: evidence from 60 demographic and health surveys. World Health Organization; 2012. https://apps.who.int/iris/bitstream/handle/10665/75429/9789241504058_eng.pdf;jsessionid=33B4B793F06D887458364DE1B36D88BA?sequence=1 .
Castle S, Askew I. Contraceptive discontinuation: reasons, challenges, and solutions. Population Council and FP2020; 2015. http://ec2-54-210-230-186.compute-1.amazonaws.com/wp-content/uploads/2015/12/FP2020_ContraceptiveDiscontinuation_SinglePage_Final_12.08.15.pdf .
Jain AK, Winfrey W. Contribution of contraceptive discontinuation to unintended births in 36 developing countries. Stud Fam Plann. 2017;48(3):269–78.
Hubacher D, Mavranezouli I, McGinn E. Unintended pregnancy in sub-Saharan Africa: magnitude of the problem and potential role of contraceptive implants to alleviate it. Contraception. 2008;78(1):73–8.
Curtis SL, Neitzel K. Contraceptive knowledge, use, and sources. In: DHS Comparative Studies No 19. Calverton, Maryland, USA: Macro International; 1996.
United Nations Department of International Economic and Social Affairs. Levels and trends of contraceptive use as assessed in 1988. United Nations Publications; 1989.
Alvergne A, Stevens R, Gurmu E. Side effects and the need for secrecy: characterising discontinuation of modern contraception and its causes in Ethiopia using mixed methods. Contracept Repro Med. 2017;2(1):24.
Ali MM, Cleland J. Oral contraceptive discontinuation and its aftermath in 19 developing countries. Contraception. 2010;81(1):22–9.
Savabi Esfahany M, Fadaei S, Yousefy A. Use of combined oral contraceptives: retrospective study in Isfahan, Islamic Republic of Iran. East Mediterr Health J. 2006;12(3–4):417–22.
CAS PubMed Google Scholar
Ojakaa D. Trends and determinants of unmet need for family planning in Kenya. In: DHS Working Papers No 56. Calverton, Maryland, USA; 2008.
Performance Monitoring and Accountability 2020 (PMA2020) Project ICRH-K. PMA 2018 Kenya Round 7 Family Planning Brief. Baltimore MD: PMA 2020, Bill and Melinda Gates Institute for Population and Reproductive Health, Johns Hopkins Bloomberg School of Public Health; 2019.
Kenya National Bureau of Statistics. Ministry of Health/Kenya, National AIDS Control Council/Kenya, Kenya Medical Research Institute, Population NCf, Development/Kenya: Kenya Demographic and Health Survey 2014. MD, USA: Rockville; 2015.
Google Scholar
Blanc AK, Curtis SL, Croft TN. Monitoring contraceptive continuation: links to fertility outcomes and quality of care. Stud Fam Plann. 2002;33(2):127–40.
PMA2020 Project ICRH-K. PMA 2018 Kenya Round 5 Family Planning Brief. Baltimore MD: PMA 2020, Bill and Melinda Gates Institute for Population and Reproductive Health, Johns Hopkins Bloomberg School of Public Health; 2017.
PMA2020 Project ICfRH-K: PMA 2018 Kenya round 6 Family Planning Brief. In. Baltimore MD: PMA 2020, Bill and Melinda Gates Institute for Population and Reproductive Health, Johns Hopkins Bloomberg School of Public Health; 2018.
Simmons RG, Sanders JN, Geist C, Gawron L, Myers K, Turok DK. Predictors of contraceptive switching and discontinuation within the first 6 months of use among highly effective reversible contraceptive initiative Salt Lake study participants. Am J Obst Gynecol. 2019;220(4):376.
Ontiri S, Mutea L, Muganda M, Mutanda P, Ajema C, Okoth S, et al. Protocol for a prospective mixed-methods longitudinal study to evaluate the dynamics of contraceptive use, discontinuation, and switching in Kenya. Reprod Health. 2019;16(1):134.
Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349–57.
Braun V, Clarke V. Using thematic analysis in psychology. Qual Res Psychol. 2006;3(2):77–101.
Corden A, Sainsbury R. Exploring ‘quality’: Research participants’ perspectives on verbatim quotations. Int J Soc Res Methodol. 2006;9(2):97–110.
Wellings K, Brima N, Sadler K, Copas AJ, McDaid L, Mercer CH, et al. Stopping and switching contraceptive methods: findings from Contessa, a prospective longitudinal study of women of reproductive age in England. Contraception. 2015;91(1):57–66.
Barden-O’Fallon J, Speizer IS, Cálix J, Rodriguez F. Contraceptive discontinuation among Honduran women who use reversible methods. Stud Fam Plann. 2011;42(1):11–20.
Azmat SK, Shaikh BT, Hameed W, Bilgrami M, Mustafa G, Ali M, et al. Rates of IUCD discontinuation and its associated factors among the clients of a social franchising network in Pakistan. BMC Women’s Health. 2012;12(1):8.
Ali MM, Sadler RK, Cleland J, Ngo TD, Shah IH. Long-term contraceptive protection discontinuation and switching behaviour. Intrauterine device (IUD) use dynamics in 14 developing countries. London: World Health Organization and Marie Stopes International; 2011.
Chebet JJ, McMahon SA, Greenspan JA, Mosha IH, Callaghan-Koru JA, Killewo J, et al. “Every method seems to have its problems”-Perspectives on side effects of hormonal contraceptives in Morogoro Region, Tanzania. BMC Women’s Health. 2015;15(1):97.
Burke H, Ambasa-Shisanya C. Qualitative study of reasons for discontinuation of injectable contraceptives among users and salient reference groups in Kenya. Af J Reprod Health. 2011;15:2.
Nanvubya A, Wanyenze RK, Kamacooko O, Nakaweesa T, Mpendo J, Kawoozo B, Matovu F, Nabukalu S, Omoding G, Kaweesi J. Barriers and facilitators of family planning use in fishing communities of Lake Victoria in Uganda. J Prim Care Community Health. 2020;11:2150132720943775.
Rademacher KH, Sergison J, Glish L, Maldonado LY, Mackenzie A, Nanda G, Yacobson I. Menstrual bleeding changes are NORMAL: proposed counseling tool to address common reasons for non-use and discontinuation of contraception. Glob Health Sci Pract. 2018;6(3):603–10.
Darroch JE, Singh S, Weissman E. Adding it up: the costs and benefits of investing in sexual and reproductive health 2014—estimation methodology. Appendix B: estimating sexual and reproductive health program and systems costs. New York: Guttmacher Institute 2016.
Trussell J. Contraceptive failure in the United States. Contraception. 2004;70(2):89–96.
Penfold S, Wendot S, Nafula I, Footman K. A qualitative study of safe abortion and post-abortion family planning service experiences of women attending private facilities in Kenya. Reprod Health. 2018;15(1):70.
Green CP, Chohen SI, Belhadj-El Ghouayel H. Male involvement in reproductive health, including family planning and sexual health. United Nations Population Fund New York; 1995.
Blumenthal PD, Voedisch A, Gemzell-Danielsson K. Strategies to prevent unintended pregnancy: increasing use of long-acting reversible contraception. Human Reprod Update. 2010;17(1):121–37.
Download references

Acknowledgements
The authors would like to acknowledge the generous contribution of time and expertise by those who participated in this study. We are grateful to Dr. Solomon Orero and Elizabeth Thompson from Jhpiego for reviewing the manuscript.
The study is funded by USAID Kenya and East Africa under Afya Halisi project, award number AID-615-A-17-00004. The funding institution did not play a role in the study design, implementation, in the writing of the manuscript, or in the decision to submit the article for publication.
Author information
Authors and affiliations.
Jhpiego Corporation, Johns Hopkins University Affiliate, Nairobi, Kenya
Susan Ontiri
Department of Health Sciences, Global Health, University Medical Centre Groningen/University of Groningen, Groningen, The Netherlands
Susan Ontiri, Regien Biesma & Jelle Stekelenburg
USAID Kenya and East Africa, Nairobi, Kenya
Lilian Mutea
School of Arts and Social Sciences, Moi University, Eldoret, Kenya
Violet Naanyu
Jhpiego Corporation, Johns Hopkins University Affiliate, Baltimore, MD, USA
Department of Obstetrics and Gynecology, Leeuwarden Medical Centre, Leeuwarden, The Netherlands
Jelle Stekelenburg
You can also search for this author in PubMed Google Scholar
Contributions
SO, LM, MK, RB and JS contributed to the design of the study. VN and SO performed data analysis. SO drafted the manuscript. All authors critically revised the manuscript and approved the final version. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Susan Ontiri .
Ethics declarations
Ethics approval and consent to participate.
Ethical approval was obtained from the Kenya Medical Research Institute’s Scientific and Ethics Review Unit, Nairobi, Kenya, (Approval number 650) and the Johns Hopkins Bloomberg School of Public Health Institutional Review Board, Baltimore, Maryland, United States of America (Approval number 00009062). Written informed consent was obtained from all study participants.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:.
Consolidated criteria for reporting qualitative research completed checklist.
Additional file 2:
Qualitative interview guides.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Ontiri, S., Mutea, L., Naanyu, V. et al. A qualitative exploration of contraceptive use and discontinuation among women with an unmet need for modern contraception in Kenya. Reprod Health 18 , 33 (2021). https://doi.org/10.1186/s12978-021-01094-y
Download citation
Received : 10 September 2020
Accepted : 31 January 2021
Published : 09 February 2021
DOI : https://doi.org/10.1186/s12978-021-01094-y
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Contraceptive discontinuation
- Contraceptive side effects
- Quality of care
- Unmet need for family planning
Reproductive Health
ISSN: 1742-4755
- General enquiries: [email protected]
- How we are governed
- Constituencies
- PMNCH Strategy, Annual Reports and Workplans
- PMNCH history
- Secretariat
- Academic, Research and Training Institutes
- Adolescents and Youth
- Donors and Foundations
- Global Financing Mechanisms
- Health-Care Professional Associations
- Inter-Governmental Organizations
- Non-Governmental Organizations
- Partner Governments
- Private Sector
- United Nations Agencies
- Working Groups
- Governance Reform
- Executive Committee
- Focus areas
- Maternal, newborn and child health (including stillbirths)
- Sexual and reproductive health and rights
- Adolescent health and well-being
- Knowledge Synthesis
- Partner Engagement
- Campaigns and Outreach

- PMNCH E-Blast
- Headlines »

- Tools and toolkits
- Publications
- Knowledge summaries
- Humanitarian and fragile settings
- All publications

- Becoming a partner
- Constituency focal points
- PMNCH Request for Proposals and Job Board
- Partner social media assets
- Digital Advocacy Hubs

PMNCH Request for Proposals and Job Board Bulletin
Section navigation.
- Our new members in 2021
- Our new members in 2022
PMNCH regularly advertises Requests for Proposals (RfPs) for various contracts related to work on maternal, newborn and child health. PMNCH often advertises Requests for proposals (RfPs) for activities and services, including consultancies.
Requests for proposals (RfPs), nominations and PMNCH jobs
Call for Nominations for PMNCH United Nations Agencies
Call for Nominations for PMNCH Private Sector Constituency
Partner opportunities
BMC: Call for papers - Improving maternal health in underserved populations
BMC: Call for submissions - Progress in reducing inequalities in health-related SDGs in the post-pandemic
WHO: Special Issue: 50 Years of Immunization—Steps Forward, submission of work
International Neonatology Association: Abstract submissions for INAC 2024
IBP Network: Call for FP/RH Program Implementation Stories
Submit your opportunity
Job and vendor announcements
- Biochemistry and Molecular Biology
- Biostatistics
- Environmental Health and Engineering
- Epidemiology
- Health Policy and Management
- Health, Behavior and Society
- International Health
- Mental Health
- Molecular Microbiology and Immunology
- Population, Family and Reproductive Health
- Program Finder
- Admissions Services
- Course Directory
- Academic Calendar
- Hybrid Campus
- Lecture Series
- Convocation
- Strategy and Development
- Implementation and Impact
- Integrity and Oversight
- In the School
- In the Field
- In Baltimore
- Resources for Practitioners
- Articles & News Releases
- In The News
- Statements & Announcements
- At a Glance
- Student Life
- Strategic Priorities
- Inclusion, Diversity, Anti-Racism, and Equity (IDARE)
- What is Public Health?
research@BSPH
The School’s research endeavors aim to improve the public’s health in the U.S. and throughout the world.
- Funding Opportunities and Support
- Faculty Innovation Award Winners
Conducting Research That Addresses Public Health Issues Worldwide
Systematic and rigorous inquiry allows us to discover the fundamental mechanisms and causes of disease and disparities. At our Office of Research ( research@BSPH), we translate that knowledge to develop, evaluate, and disseminate treatment and prevention strategies and inform public health practice. Research along this entire spectrum represents a fundamental mission of the Johns Hopkins Bloomberg School of Public Health.
From laboratories at Baltimore’s Wolfe Street building, to Bangladesh maternity wards in densely packed neighborhoods, to field studies in rural Botswana, Bloomberg School faculty lead research that directly addresses the most critical public health issues worldwide. Research spans from molecules to societies and relies on methodologies as diverse as bench science and epidemiology. That research is translated into impact, from discovering ways to eliminate malaria, increase healthy behavior, reduce the toll of chronic disease, improve the health of mothers and infants, or change the biology of aging.
120+ countries
engaged in research activity by BSPH faculty and teams.
of all federal grants and contracts awarded to schools of public health are awarded to BSPH.
citations on publications where BSPH was listed in the authors' affiliation in 2019-2023.
publications where BSPH was listed in the authors' affiliation in 2019-2023.
Departments
Our 10 departments offer faculty and students the flexibility to focus on a variety of public health disciplines
Centers and Institutes Directory
Our 80+ Centers and Institutes provide a unique combination of breadth and depth, and rich opportunities for collaboration
Institutional Review Board (IRB)
The Institutional Review Board (IRB) oversees two IRBs registered with the U.S. Office of Human Research Protections, IRB X and IRB FC, which meet weekly to review human subjects research applications for Bloomberg School faculty and students
Generosity helps our community think outside the traditional boundaries of public health, working across disciplines and industries, to translate research into innovative health interventions and practices
Introducing the research@BSPH Ecosystem
The research@BSPH ecosystem aims to foster an interdependent sense of community among faculty researchers, their research teams, administration, and staff that leverages knowledge and develops shared responses to challenges. The ultimate goal is to work collectively to reduce administrative and bureaucratic barriers related to conducting experiments, recruiting participants, analyzing data, hiring staff, and more, so that faculty can focus on their core academic pursuits.

Research at the Bloomberg School is a team sport.
In order to provide extensive guidance, infrastructure, and support in pursuit of its research mission, research@BSPH employs three core areas: strategy and development, implementation and impact, and integrity and oversight. Our exceptional research teams comprised of faculty, postdoctoral fellows, students, and committed staff are united in our collaborative, collegial, and entrepreneurial approach to problem solving. T he Bloomberg School ensures that our research is accomplished according to the highest ethical standards and complies with all regulatory requirements. In addition to our institutional review board (IRB) which provides oversight for human subjects research, basic science studies employee techniques to ensure the reproducibility of research.
Research@BSPH in the News
Four bloomberg school faculty elected to national academy of medicine.
Considered one of the highest honors in the fields of health and medicine, NAM membership recognizes outstanding professional achievements and commitment to service.
The Maryland Maternal Health Innovation Program Grant Renewed with Johns Hopkins
Lerner center for public health advocacy announces inaugural sommer klag advocacy impact award winners.
Bloomberg School faculty Nadia Akseer and Cass Crifasi selected winners at Advocacy Impact Awards Pitch Competition

An official website of the United States government
Here’s how you know
Official websites use .gov A .gov website belongs to an official government organization in the United States.
Secure .gov websites use HTTPS A lock ( Lock A locked padlock ) or https:// means you’ve safely connected to the .gov website. Share sensitive information only on official, secure websites.
HHS FY 2025 Budget in Brief
Topics on this page: HHS Budget in Brief | Budget Justifications | Office of the Secretary Staff Divisions | Operating Division Requests | GAO-IG Act | Offices to Contact | Previous Budgets FY 2024 – FY2020
The President’s Fiscal Year (FY) 2025 Budget supports the Department of Health and Human Services’ (HHS) mission to promote the health and well-being of all Americans. HHS proposes $130.7 billion in discretionary and $1.7 trillion in mandatory proposed budget authority for FY 2025.
This budget illustrates HHS’s commitment to support American families, improve behavioral health, and ensure the nation’s readiness for the next public health crisis. The budget works to ensure all Americans have access to affordable healthcare; improve maternal and reproductive health outcomes; strengthen early care and education; address the needs of Indian Country; and advance scientific innovation.
This budget supports HHS’s mission by investing in critical program operations and infrastructure. Serving the American people is fundamental to meeting HHS’s mission, and in FY 2025, HHS will also support multiple customer experience efforts to improve HHS's service delivery.
At the time of final preparation of the budget, Congress has not yet set final discretionary funding levels for FY 2024. As a result, the budget shows discretionary funding comparisons to FY 2023, and mandatory funding comparisons to FY 2024 current law levels.
Read the full FY 2025 Budget in Brief *
Budget Justifications to Congress
NOTE: IT Resource Statement:
HHS certifies that the HHS Chief Information Officer (CIO) has reviewed and had input in approving information technology (IT) Investments included in the below budget request documents. Furthermore, both the HHS Chief Financial Officer (CFO) and HHS CIO have had a role in reviewing planned IT support for major programs and significant increases and decreases in IT resources as reflected in this budget. Additionally, with respect to Federal Information Technology Acquisition Reform Act (FITARA) implementation, the Agency has developed and implemented its plan to ensure that all common baseline FITARA responsibilities are in place. Finally, HHS confirms that all HHS components are utilizing incremental development practices as appropriate across their IT investment portfolio.
NOTE: 21st Century Integrated Digital Experience Act (IDEA) Modernization
Office of the Secretary Staff Divisions (Consolidated):
- General Departmental Management *
- Office of Inspector General **
Operating Divisions Budget Requests:
- Administration for Children and Families (ACF)
- Administration for Community Living (ACL)
- Agency for Healthcare Research and Quality (AHRQ)
- Advanced Research Projects Agency for Health (ARPA-H)
- Administration for Strategic Preparedness and Response (ASPR)
- Agency for Toxic Substances and Disease Registry (ATSDR)
- Centers for Disease Control and Prevention (CDC)
- Centers for Medicare & Medicaid Services (CMS)
- Food and Drug Administration (FDA)
- Health Resources and Services Administration (HRSA)
- Indian Health Service (IHS)
- National Institutes of Health (NIH)
- Substance use And Mental Health Services Administration (SAMHSA)
- HHS GAO-IG Act Report
Offices to Contact
- Assistant Secretary for Financial Resources
- Office of Budget
HHS Budget – Previous Years
- FY 2024 Budget
- FY 2023 Budget
- FY 2022 Budget
- FY 2021 Budget
- FY 2020 Budget
*This content is in the process of Section 508 remediation. If you need immediate assistance accessing this content, please submit a request to [email protected] .
**This content is in the process of Section 508 remediation. If you need immediate assistance accessing this content, please submit a request to [email protected] .
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List

Effect of Intermittent Fasting on Reproductive Hormone Levels in Females and Males: A Review of Human Trials
Associated data.
Not applicable.
Intermittent fasting is a popular diet for weight loss, but concerns have been raised regarding the effects of fasting on the reproductive health of women and men. Accordingly, we conducted this literature review to clarify the effects of fasting on reproductive hormone levels in humans. Our results suggest that intermittent fasting decreases androgen markers (i.e., testosterone and the free androgen index (FAI)) while increasing sex hormone-binding globulin (SHBG) levels in premenopausal females with obesity. This effect was more likely to occur when food consumption was confined to earlier in the day (eating all food before 4 pm). In contrast, fasting did not have any effect on estrogen, gonadotropins, or prolactin levels in women. As for men, intermittent fasting reduced testosterone levels in lean, physically active, young males, but it did not affect SHBG concentrations. Interestingly, muscle mass and muscular strength were not negatively affected by these reductions in testosterone. In interpreting these findings, it is important to note that very few studies have been conducted on this topic. Thus, it is difficult to draw solid conclusions at present. From the limited data presented here, it is possible that intermittent fasting may decrease androgen markers in both genders. If this is the case, these results would have varied health implications. On the one hand, fasting may prove to be a valuable tool for treating hyperandrogenism in females with polycystic ovarian syndrome (PCOS) by improving menstruation and fertility. On the other hand, fasting may be shown to decrease androgens among males, which could negatively affect metabolic health and libido. More research is warranted to confirm these preliminary findings.
1. Introduction
Intermittent fasting has gained tremendous popularity over the last decade as a weight loss regimen [ 1 , 2 ]. Intermittent fasting is an umbrella term for three different diets: alternate day fasting (ADF), the 5:2 diet, and time-restricted eating (TRE). ADF involves a “feast day”, where individuals eat ad libitum, alternated with a “fast day”, where participants can choose to consume only water or consume ∼25% of energy needs. The 5:2 diet, on the other hand, is a modified version of ADF that involves 5 feast days and 2 fast days per week. Finally, TRE involves confining the eating window to a specified number of hours per day (usually 4 to 10 h) and fasting with zero-calorie beverages for the remaining hours [ 1 , 2 ]. Accumulating evidence indicates that these various intermittent fasting regimens are effective for decreasing body weight and improving insulin sensitivity, blood pressure, and markers of oxidative stress in adults with obesity [ 1 , 3 , 4 , 5 ].
Nevertheless, concerns have been raised regarding the effects of fasting on the reproductive health of women and men. For instance, some women are skeptical about starting intermittent fasting because they believe it may negatively affect levels of estrogen and other reproductive hormones, leading to menstrual cycle irregularities and fertility issues. These concerns largely stem from the findings of one rodent study by Kumar et al. [ 6 ]. This study [ 6 ] is heavily referenced in the popular media and is often used as evidence to show that fasting is disruptive to female reproduction. In their experiment [ 6 ], young rats underwent 24 h of water fasting every other day for 12 weeks. By the end of the trial, serum estradiol increased, while luteinizing hormone (LH) levels decreased versus ad libitum fed controls. Negative changes in estrous cyclicity (i.e., menstrual cycle) were also observed. While these findings are indeed concerning, it should be noted that the female rats were very young (3 months old), which corresponds to a human aged 9 years old [ 7 ]. Intermittent fasting is not recommended for children under the age of 12, since it has the potential to negatively impact puberty and growth [ 2 ]. Thus, data from the Kumar et al. study [ 6 ], though valuable to the field, do not provide evidence for how fasting may impact reproductive hormone levels in adult females.
Some men are also wary about starting intermittent fasting, as they believe it may alter testosterone levels. These concerns come from a study by Moro et al. [ 8 ], where young lean males participated in an 8 h TRE intervention (all food consumed between 12 to 8 p.m.) combined with resistance training. After 8 weeks of intervention, reductions in free and total testosterone concentrations were observed. Interestingly, the decreases in the anabolic hormone, testosterone, did not lead to any deleterious body composition changes or compromises in muscular strength. These findings highlight the need for more research to be conducted in this field.
To date, the effects of intermittent fasting on reproductive hormone levels in humans remain largely unknown, since no reviews of the literature have been performed. In view of this research gap, we conducted this review to further clarify the effects of intermittent fasting on sex hormone levels in adult female and male participants.
2. Methods—Human Trial Selection
A PubMed, CINAHL, EBSCOhost, and Cochrane Library search was conducted using the following key words: “intermittent fasting”, “meal timing”, “meal frequency”, “delayed meal”, “intermittent energy restriction”, “intermittent calorie restriction”, “alternate day fasting”, “5:2 diet”, “time restricted eating”, “time restricted feeding”, “testosterone”, “estrogen”, “estradiol”, “progesterone”, “prolactin”, “DHEA”, “follicle-stimulating hormone”, “luteinizing hormone”, “androgen”, “sex hormone binding globulin”, “sex hormone”, and “free androgen index”. The inclusion criteria for research articles were as follows: (1) randomized controlled trials and nonrandomized trials; (2) adult male and female participants (>18 years); (3) endpoints that included changes in body weight and at least one sex hormone. The following exclusion criteria were applied: (1) cohort and observational studies; (2) fasting performed as a religious practice (e.g., Ramadan or Seventh Day Adventist); (3) trial durations of less than 1 week; (4) studies that combined data for males and females. Our search retrieved 5 human trials of TRE, [ 8 , 9 , 10 , 11 , 12 ] one human trial of the 5:2 diet, [ 13 ], and one study [ 14 ] that examined the effect of meal timing on reproductive hormone concentrations. The results from these trials were separated for females ( Table 1 ) and males ( Table 2 ). We were not able to find any studies on ADF that examined sex hormones; therefore, this form of fasting was not included in this review.
Females: effect of intermittent fasting on reproductive hormones concentrations.
| Study Design | % Change from Baseline | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reference | Subjects | Duration (Weeks) | Interventions | BW | FM | FFM | Estradiol | Testosterone, AE | SHBG | FAI | DHEA | LH FSH PRL |
| Li 2021 [ ] | = 18 Females Age: 18–31 y Overweight Obese Premenopausal PCOS | 5 | 1. 8 h TRE (8 a.m.–4 p.m.) | 1. ↓2% * | -- | -- | -- | Total T: 1. ↓9% * | 1. ↑2% * | 1. ↓26% * | -- | 1. LH: ∅ FSH: ∅ |
| Harvie 2011 [ ] | = 107 Females Age: 30–45 y Overweight Obese Premenopausal | 24 | 1. 5:2 Diet: Fast day (500 kcal), Feast day (ad libitum) 2. CR (1500 kcal/d) | 1. ↓7% * 2. ↓5% * | 1. ↓ * 2. ↓ * | 1. ↓* 2. ↓ * | -- | Free T: 1. ∅ 2. ∅ AE: 1. ∅ 2. ∅ | 1. ↑14% * 2. ↑6%* | 1. ↓6% * 2. ↓10% * | 1. ∅ 2. ↓6% * | PRL: 1. ∅ 2. ∅ |
| Jakubo-wicz 2013 [ ] | = 60 Females Age: 25–39 y Overweight Normal weight Premenopausal PCOS | 12 | 1. >50% of daily calories consumed at breakfast 2. >50% of daily calories consumed at dinner | 1. ∅ 2. ∅ | -- | -- | 1. ∅ 2.↑35% * | Free T: 1. ↓50% *† 2. ∅ Total T: 1. ↓47% *† 2. ∅ AE: 1. ↓34% *† 2. ∅ | 1.↑% *† 2. ∅ | 1.↓% *† 2. ∅ | 1.↓% *† 2. ∅ | -- |
--: Not measured; ∅: nonsignificant change, ↓: decrease; ↑: increase. * p < 0.05, significantly different from baseline (within a group effect). † p < 0.05, significantly different from the control or comparison group (between group effect). AE: androstenedione; BW: body weight; CR: calorie restriction; DHEA-S: dehydroepiandrosterone sulfate; FAI: free androgen index (100 × (total testosterone/SHBG)); FFM: fat-free mass; FM: fat mass; FSH: follicle-stimulating hormone; LH: luteinizing hormone; PRL: prolactin; RT: randomized trial; SHBG: sex hormone-binding globulin; T: testosterone, TRE: time-restricted eating (prescribed eating window shown in parentheses); y: years.
Males: effect of intermittent fasting on reproductive hormone concentrations.
| Study Design | % Change from Baseline | |||||||
|---|---|---|---|---|---|---|---|---|
| Reference | Subjects | Duration (Weeks) | Interventions | BW | FM | FFM | Testosterone | SHBG |
| Stratton 2020 [ ] | = 26 Males Age: 18–35 y Physically active | 4 | 1.8 h TRE + CR25% + Resistance training 3×/week 2. CR 25% + Resistance training 3×/week | 1. ↓1% * 2. ↓2% * | 1. ↓9% * 2. ↓9% * | 1. ∅ 2. ∅ | Total T: 1. ↓1% * 2. ↓1% * | -- |
| Moro 2020 [ ] | = 16 Males Age: 19 ± 2 y Elite cyclists | 4 | 1. 8 h TRE (10 a.m.–7 p.m.) 2. Usual diet (7 a.m.–9 p.m.) | 1. ↓2% *† 2. ∅ | 1. ∅ 2. ∅ | 1. ∅ 2. ∅ | Free T: 1. ↓27% *† 2. ↓8%† | 1. ∅ 2. ∅ |
| Moro 2016 [ ] | = 34 Males Age: 29 ± 4 y Resistance trained | 8 | 1. 8 h TRE (12 p.m.–8 p.m.) + Resistance training 3×/week 2. Usual diet (8 a.m.–8 p.m.) + Resistance training 3×/week | 1. ↓ * 2. ∅ | 1. ↓15% *† 2. ∅ | 1. ∅ 2. ∅ | Total T: 1. ↓21% *† 2. ∅ | -- |
| Moro 2021 [ ] | = 20 Males Age: 29 ± 4 y Resistance trained | 44 | 1. 8 h TRE (1 p.m.–8 p.m.) + Resistance training 3×/week 2. Usual diet (8 a.m.–8 p.m.) + Resistance training 3×/week | 1. ↓3% *† 2. ↑3% *† | 1. ↓12% * 2. ∅ | 1. ∅ 2. ↑3% * | Total T: 1. ↓17% * 2. ∅ | -- |
--: Not measured; ∅: nonsignificant change. ↓: decrease; ↑: increase. * p < 0.05, significantly different from baseline (within group effect). † p < 0.05, significantly different from the control or comparison group (between group effect). BW: body weight; FFM: fat-free mass; FM: fat mass; RT: randomized trial; RCT: randomized control trial; SHBG: sex hormone-binding globulin; T: testosterone; TRE: time-restricted eating (prescribed eating window shown in parentheses).
2.1. Females: Effects of Intermittent Fasting on Reproductive Hormone Concentrations
2.1.1. estradiol.
Estradiol is a naturally occurring form of estrogen [ 15 ]. It is the main female reproductive hormone and is involved in the development and maintenance of female reproductive tissues and the regulation of the menstrual cycle [ 15 ]. Women with overweight and obesity have higher levels of estrogens relative to their normal-weight counterparts [ 16 ]. An increase in estrogens derived from excess adiposity is linked to adverse health outcomes such as polycystic ovarian syndrome (PCOS), anovulation, and increased breast cancer risk [ 17 , 18 , 19 ]. Weight loss interventions have been shown to reduce estrogen levels among females with obesity [ 20 ].
To date, only one trial [ 14 ] has measured the effect of meal timing on estradiol levels in women ( Table 1 ). Jakubowicz et al. [ 14 ] compared the effect of eating >50% of calories at dinner versus eating >50% of calories at breakfast in females with PCOS. After 12 weeks, estradiol significantly increased among females with PCOS when participants ate >50% of daily calories at dinner [ 14 ]. This result suggests that eating a large amount of food later in the day may augment estrogen levels in women with PCOS. Elevated serum androgens are converted to estrogens in adipose tissue, leading to excess estrogen production in females with PCOS and obesity. The increased estrogen production impairs the function of the hypothalamic–pituitary–gonadal axis. Thus, excess of estrogen and androgen are the primary causes of anovulation in PCOS patients [ 19 ]. Taken together, shifting calorie intake to earlier in the day may be preferred for females with PCOS to avoid further increases in estrogen levels.
2.1.2. Androgens (Testosterone, DHEA-S, Androstenedione, and FAI)
Hyperandrogenism is a medical condition characterized by high levels of androgens, i.e., testosterone, dehydroepiandrosterone-sulfate (DHEA-S), and androstenedione. In females, hyperandrogenism is portrayed by hirsutism (i.e., excessive hair growth), seborrhea (scaly patches on the body and scalp), and disorders in the menstrual cycle [ 21 ]. Studies have reported that hyperandrogenism promotes insulin resistance and visceral adiposity among females by decreasing whole-body glucose uptake [ 22 , 23 ]. In females with PCOS and obesity, weight loss has been shown to decrease testosterone and androstenedione levels while increasing SHBG concentrations [ 24 , 25 ]. Abnormal androgen status is measured by the free androgen index (FAI). The FAI is a ratio that is calculated by dividing total testosterone by sex hormone-binding globulin (SHBG) and then multiplying by 100.
Three studies [ 9 , 13 , 14 ] have examined the effect of intermittent fasting on androgen markers in females ( Table 1 ). In a study by Harvie et al. [ 13 ], premenopausal women with obesity followed a 5:2 diet where they fasted with 500 kcal two days per week. After 24 weeks of 5:2, the FAI significantly decreased, with a 7% weight loss versus baseline. DHEA-S, testosterone, and androstenedione, on the other hand, remained unchanged [ 13 ]. In addition, two studies [ 9 , 14 ] examined the effects of fasting on androgens in women with PCOS. Li et al. [ 9 ] conducted an 8 h TRE trial, where young women with PCOS and obesity ate all of their energy needs early in the day (between 8 am and 4 pm) and fasted with water for the rest of the day for 5 weeks. This early 8 h TRE intervention significantly decreased body weight by 2%, along with FAI and total testosterone levels. Complementary to these findings, Jakubowicz et al. [ 14 ] compared the effect of eating >50% of calories at dinner versus eating >50% of calories at breakfast in females with PCOS. The results showed that FAI, DHEA-S, and androstenedione decreased significantly in the breakfast group relative to the dinner group. These changes occurred without weight loss. In these trials [ 9 , 13 , 14 ], changes in androgen markers were also accompanied by reductions in body weight, inflammation, and insulin resistance, further illustrating the link between hyperandrogenism and metabolic disturbances. Altogether, these findings suggest that fasting can significantly decrease androgen markers in premenopausal females and those with PCOS, especially when calories are consumed earlier in the day.
2.1.3. SHBG
Sex hormone-binding globulin (SHBG) is a hepatocyte-produced glycoprotein. The principal function of SHBG is to transport testosterone and estradiol to target tissues. Thus, the bioavailability of these reproductive hormones is influenced by circulating levels of SHBG [ 26 ]. Observational studies show that low levels of SHBG are associated with an increased incidence in insulin resistance and type 2 diabetes, independent of sex hormone concentrations [ 27 ]. Moreover, low levels of SHBG are frequently found in females with PCOS and contribute to hyperandrogenic symptoms such as hirsutism, acne, and androgenic alopecia [ 28 , 29 , 30 ]. Weight loss and has been shown to increase SHBG and improve insulin sensitivity in women with obesity and PCOS [ 31 , 32 ].
Three trials [ 9 , 13 , 14 ] examined how fasting impacts circulating concentrations of SHBG in females ( Table 1 ). Li et al. [ 9 ] showed that concentrations of SHBG significantly increased, with 2% weight loss after 5 weeks of 8 h TRE in females with PCOS. The study by Harvie et al. [ 13 ] also demonstrated a significant increase in circulating SHBG levels, with a 7% weight loss after 24 weeks of the 5:2 diet in premenopausal women with obesity. Moreover, in the study by Jakubowicz et al. [ 14 ], SHBG levels increased when females with PCOS ate >50% of their calories in the morning compared to eating those calories in the evening. Interestingly, these improvements in SHBG occurred despite no change in body weight [ 14 ]. These preliminary findings suggest that intermittent fasting regimens may produce beneficial increases in SHBG concentrations in premenopausal females and those with PCOS, particularly when most of the food is consumed in the morning or afternoon.
2.1.4. Gonadotropins
Gonadotropins are peptide hormones that regulate ovarian function and are essential for normal growth, sexual development, and reproduction [ 33 ]. Human gonadotropins include follicle-stimulating hormone (FSH) and luteinizing hormone (LH), which are made in the pituitary gland [ 33 ]. Previous studies have shown that weight loss reduces the LH/FSH ratio, with an FSH predominance favoring the maturation of the ovarian follicle (folliculogenesis) [ 34 ].
Changes in gonadotropins during fasting have only been assessed in one clinical trial to date [ 9 ] ( Table 1 ). In this trial by Li et al. [ 9 ], young women with obesity and PCOS followed an early 8 h TRE regimen for 5 weeks. At the conclusion of the study, LH and FSH remained unchanged, even though these participants lost a small amount of weight (2% from baseline) and fat mass (2.4 kg from baseline). Visceral fat also decreased, but the skeletal muscle remained unchanged. It is possible that the degree of weight loss and fat mass loss was not sufficient to modulate the LH and FSH concentrations.
2.1.5. Prolactin
Prolactin is a hormone responsible for milk production and mammary gland development [ 35 ]. Accumulating evidence suggests that weight loss via dietary interventions does not significantly affect prolactin levels [ 36 , 37 ]. Prolactin concentrations have only been assessed in one study on intermittent fasting [ 13 ]. In this trial by Harvie et al. [ 13 ], prolactin levels remained unchanged after 24 weeks of the 5:2 diet in premenopausal women with overweight and obesity. These results, although very limited, suggest that intermittent fasting may be safe for lactating females. In support of this, a recent trial by Gray et al. [ 38 ] studied the effect of the 5:2 diet on weight loss and gestational diabetes risk reduction in breastfeeding women. The study showed that intermittent fasting was safe in this population group, well tolerated, and lead to no adverse changes in milk production. These findings warrant confirmation by a study that specifically examines the safety and efficacy of intermittent fasting in breastfeeding women.
2.1.6. Females: Summary of Findings
These preliminary findings suggest that fasting generally decreases androgens (i.e., testosterone and FAI) while increasing SHBG in premenopausal females with obesity. These results offer promise for the use of intermittent fasting in the treatment of hyperandrogenic conditions such as PCOS. Though it is important to note that these results were generally only observed when food consumption was limited to earlier in the day. Thus, premenopausal women may need to finish eating by 4 pm each day to observe these benefits in androgen markers. On the other hand, fasting does not appear to have any effect on other reproductive hormones such as estrogen, gonadotropins, and prolactin. More research is needed to confirm these findings.
2.2. Males: Effects of Intermittent Fasting on Reproductive Hormone Concentrations
2.2.1. testosterone.
Testosterone is the major androgenic steroid hormone in adult males and is responsible for maintaining sperm production, libido, and sexual efficacy [ 39 ]. Another key role of testosterone is to stimulate muscle protein synthesis, thereby increasing muscle mass.
The effects of fasting on testosterone levels have been examined in four clinical trials to date [ 8 , 10 , 11 , 12 ] ( Table 2 ). All studies employed 8 h TRE as the fasting intervention, and most combined TRE with resistance training (three times per week). Each trial was conducted in physically active, lean, young men. The findings revealed that TRE alone and combined with resistance training consistently reduced total testosterone levels [ 8 , 10 , 12 ] and free testosterone [ 11 ] after 4 to 44 weeks of intervention. The reductions in testosterone did not appear to be related to the duration of intervention, as shorter trials produced similar changes in testosterone as longer trials [ 8 , 10 , 11 , 12 ]. Mild weight loss was noted in each trial (1–3% from baseline) [ 8 , 10 , 11 , 12 ], and the degree of weight loss did not seem to be related to the magnitude of the testosterone reduction. Body composition changes were also evaluated. In each trial, fat-free mass remained unchanged, while fat mass was reduced [ 8 , 10 , 11 , 12 ]. Interestingly, the decreases in the anabolic hormone, testosterone, did not lead to any deleterious body composition changes or compromises in muscular strength.
2.2.2. SHBG
SHBG is a glycoprotein that serves as a major carrier of testosterone in the circulation—carrying 40–45% of bound testosterone—and, thus, serum testosterone levels are higher when SHBG levels are higher [ 40 , 41 , 42 ]. Obesity can directly contribute to lower testosterone levels in males by reducing levels of SHBG [ 43 , 44 ].
Only one fasting study [ 11 ] has measured SHBG levels in male participants. After 4 weeks of 8 h TRE alone, circulating concentrations of SHBG remained unchanged, but free testosterone was reduced [ 11 ]. Less free testosterone would suggest that more testosterone is bound by carriers, but surprisingly, no subsequent increases in SHBG were observed [ 11 ]. This finding suggests that total testosterone may have decreased or that testosterone became bound to other carriers such as albumin [ 40 ]. The short time frame of the study (4 weeks) may have also prevented meaningful changes in SHBG from being observed. Taken together, TRE can decrease testosterone levels among healthy, active males, without affecting SHBG levels.
2.3. Males: Summary of Findings
These findings suggest that TRE reduces free and total testosterone levels in lean, physically active, young men. SHBG, however, does not seem to change with TRE, though the findings are very limited. Interestingly, muscle mass and muscular strength were not negatively affected by the reduction in circulating testosterone levels.
3. Intermittent Fasting, the Gut Microbiome, and Sex Hormones
Studies have shown that alterations in the gut microbiome can significantly affect reproductive hormones [ 45 ]. Improving abnormal microbiomes may lead to better reproductive health outcomes among pre- and postmenopausal women [ 46 ]. Clinical and preclinical data have shown that intermittent fasting can improve the composition and diversity of the gut microflora [ 47 , 48 ]. Periods of fasting have also been shown to reduce gut permeability leading to blunted postprandial endotoxemia and systemic inflammation, which are typically elevated in obesity [ 47 , 48 ]. Various members of the microbial community in the gastrointestinal tract can utilize endogenous (host) substrates during fasting, resulting in the production of metabolites beneficial to the host such as butyrate, acetate, and mucin stimulants [ 48 , 49 ]. Thus, it is possible that the effect of intermittent fasting on reproductive hormones is mediated by changes in the gut microbiome.
4. Circadian Rhythmicity and Sex Hormones
It has been proposed that intermittent fasting impacts sex hormone levels via improved alignment of circadian rhythms [ 14 , 50 ]. Hormone production and release are, in part, controlled by circadian rhythms which are, in turn, influenced by daily feeding–fasting cycles [ 50 ]. It has been suggested that TRE shortens the daily eating window to better align with circadian biology, which may beneficially affect hormone levels [ 14 , 50 ]. As demonstrated by Jakubowicz et al. [ 14 ], there may be a benefit to timing caloric intake earlier in the day vs. later in regard to testosterone levels in women with PCOS. While these findings offer promise for the use of early TRE in improving certain sex hormone concentrations, further research is needed before solid conclusions can be reached.
5. Directions for Future Research
The evidence in this area is still very limited. Future studies in women should involve perimenopausal and postmenopausal females, since no studies have been performed in these groups of women to date. It will also be of interest to further explore the effect of different fasting regimens in women with PCOS. Given that TRE shows promise in treating hyperandrogenism in females with this condition, large well-powered RCTs will be needed to evaluate if fasting is indeed a viable treatment option. Future studies should also examine how other sex hormones in females respond to fasting such as progesterone and estrone. In males, it will be important to include those who are overweight or obese, as current studies have only involved athletic, healthy, lean males. Trials with longer duration (24–52 weeks) are also needed.
6. Limitations to the Current Body of Evidence
There are several limitations to the current body of evidence. First, very few studies have examined how intermittent fasting impacts sex hormones. This should be taken into consideration when interpreting the present findings. Second, these trials all had small sample sizes ( n = 16–107) and measured reproductive hormones as secondary exploratory outcome measures. Thus, it is highly likely that none of these trials were adequately powered to detect statistically significant changes in any hormonal parameter. Third, many trials did not include a control group in their design. Thus, it is difficult to confirm if these results are due to the fasting intervention instead of other extraneous variables. Fourth, most of these trials were quite short; therefore, the long-term effects of intermittent fasting on reproductive hormones are still not known. Fifth, studies in women to date are limited to premenopausal females, while studies in men are limited to athletic, healthy, lean males. There are no studies in postmenopausal women or overweight/obese men, which greatly decreases the generalizability of our findings. Sixth, it is unclear if the day of the menstrual cycle was recorded and standardized for all the studies involving premenopausal females. Levels of LH, FSH, and estrogen vary considerably according to the day of menstruation and, thus, should be controlled for in future studies in this area.
7. Conclusions
In interpreting these findings, it is important to note that very few studies have been conducted in this topic area. Thus, it is difficult to draw solid conclusions at present. From the limited data presented here, it is possible that intermittent fasting may decrease androgen markers in both genders. If these findings are confirmed by future research, these results would have different health consequences for females and males. On the one hand, fasting may prove to be a valuable tool for treating hyperandrogenism in females with PCOS by improving menstruation, fertility, and quality of life. On the other hand, fasting may be shown to decrease androgens, which would be less desirable among males. Low testosterone levels can negatively affect metabolic health, muscle mass synthesis, and libido in males. Interestingly, there were no adverse changes in fat-free mass in response to this reduction in androgens. All other reproductive hormones remained unchanged for both genders in these short-term trials. While these findings provide some preliminary evidence, these data require confirmation by large-scale, well-powered RCTs designed to specifically examine how intermittent fasting impacts reproductive hormone levels in various population groups.
Funding Statement
This research received no external funding.
Author Contributions
Conceptualization, S.C. (Sofia Cienfuegos) and S.C. (Sarah Corapi); methodology, S.C. (Sofia Cienfuegos) and S.C. (Sarah Corapi); formal analysis, S.C. (Sofia Cienfuegos), S.C. (Sarah Corapi), K.G., M.E., F.K., S.L. and V.P.; writing—original draft preparation, S.C. (Sofia Cienfuegos), S.C. (Sarah Corapi), K.G., M.E., F.K., S.L., V.P. and K.A.V.; writing—review and editing, S.C. (Sofia Cienfuegos), S.C. (Sarah Corapi), K.G., M.E., F.K., S.L., V.P. and K.A.V. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Informed consent statement, data availability statement, conflicts of interest.
The authors declare no conflict of interest.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appointments at Mayo Clinic
- Stress management
- Chronic stress puts your health at risk
Chronic stress can wreak havoc on your mind and body. Take steps to control your stress.
Your body is made to react to stress in ways meant to protect you against threats from predators and other aggressors. Such threats are rare today. But that doesn't mean that life is free of stress.
Instead, you likely face many demands each day. For example, you may take on a huge workload, pay bills or take care of your family. Your body treats these everyday tasks as threats. Because of this, you may feel as if you're always under attack. But you can fight back. You don't have to let stress control your life.
Understanding the natural stress response
When you face a perceived threat, a tiny region at the brain's base, called the hypothalamus, sets off an alarm system in the body. An example of a perceived threat is a large dog barking at you during your morning walk. Through nerve and hormonal signals, this system prompts the adrenal glands, found atop the kidneys, to release a surge of hormones, such as adrenaline and cortisol.
Adrenaline makes the heart beat faster, causes blood pressure to go up and gives you more energy. Cortisol, the primary stress hormone, increases sugar, also called glucose, in the bloodstream, enhances the brain's use of glucose and increases the availability of substances in the body that repair tissues.
Cortisol also slows functions that would be nonessential or harmful in a fight-or-flight situation. It changes immune system responses and suppresses the digestive system, the reproductive system and growth processes. This complex natural alarm system also communicates with the brain regions that control mood, motivation and fear.
When the natural stress response goes wild
The body's stress response system is usually self-limiting. Once a perceived threat has passed, hormones return to typical levels. As adrenaline and cortisol levels drop, your heart rate and blood pressure return to typical levels. Other systems go back to their regular activities.
But when stressors are always present and you always feel under attack, that fight-or-flight reaction stays turned on.
The long-term activation of the stress response system and too much exposure to cortisol and other stress hormones can disrupt almost all the body's processes. This puts you at higher risk of many health problems, including:
- Depression.
- Digestive problems.
- Muscle tension and pain.
- Heart disease, heart attack, high blood pressure and stroke.
- Sleep problems.
- Weight gain.
- Problems with memory and focus.
That's why it's so important to learn healthy ways to cope with your life stressors.
Why you react to life stressors the way you do
Your reaction to a potentially stressful event is different from everyone else's. How you react to your life stressors is affected by such factors as:
- Genetics. The genes that control the stress response keep most people at a fairly steady emotional level, only sometimes priming the body for fight or flight. More active or less active stress responses may stem from slight differences in these genes.
- Life experiences. Strong stress reactions sometimes can be traced to traumatic events. People who were neglected or abused as children tend to be especially at risk of experiencing high stress. The same is true of airplane crash survivors, people in the military, police officers and firefighters, and people who have experienced violent crime.
You may have some friends who seem relaxed about almost everything. And you may have other friends who react strongly to the slightest stress. Most people react to life stressors somewhere between those extremes.
Learning to react to stress in a healthy way
Stressful events are facts of life. And you may not be able to change your current situation. But you can take steps to manage the impact these events have on you.
You can learn to identify what causes you stress. And you can learn how to take care of yourself physically and emotionally in the face of stressful situations.
Try these stress management tips:
- Eat a healthy diet and get regular exercise. Get plenty of sleep too.
- Do relaxation exercises such as yoga, deep breathing, massage or meditation.
- Keep a journal. Write about your thoughts or what you're grateful for in your life.
- Take time for hobbies, such as reading or listening to music. Or watch your favorite show or movie.
- Foster healthy friendships and talk with friends and family.
- Have a sense of humor. Find ways to include humor and laughter in your life, such as watching funny movies or looking at joke websites.
- Volunteer in your community.
- Organize and focus on what you need to get done at home and work and remove tasks that aren't needed.
- Seek professional counseling. A counselor can help you learn specific coping skills to manage stress.
Stay away from unhealthy ways of managing your stress, such as using alcohol, tobacco, drugs or excess food. If you're worried that your use of these products has gone up or changed due to stress, talk to your health care provider.
There are many rewards for learning to manage stress. For example, you can have peace of mind, fewer stressors and less anxiety, a better quality of life, improvement in conditions such as high blood pressure, better self-control and focus, and better relationships. And it might even lead to a longer, healthier life.
There is a problem with information submitted for this request. Review/update the information highlighted below and resubmit the form.
From Mayo Clinic to your inbox
Sign up for free and stay up to date on research advancements, health tips, current health topics, and expertise on managing health. Click here for an email preview.
Error Email field is required
Error Include a valid email address
To provide you with the most relevant and helpful information, and understand which information is beneficial, we may combine your email and website usage information with other information we have about you. If you are a Mayo Clinic patient, this could include protected health information. If we combine this information with your protected health information, we will treat all of that information as protected health information and will only use or disclose that information as set forth in our notice of privacy practices. You may opt-out of email communications at any time by clicking on the unsubscribe link in the e-mail.
Thank you for subscribing!
You'll soon start receiving the latest Mayo Clinic health information you requested in your inbox.
Sorry something went wrong with your subscription
Please, try again in a couple of minutes
- How stress affects your health. American Psychological Association. https://www.apa.org/topics/stress/health. Accessed March 19, 2021.
- Stress effects on the body. American Psychological Association. https://www.apa.org/topics/stress/body. Accessed March 19, 2021.
- Lower stress: How does stress affect the body? American Heart Association. https://www.heart.org/en/healthy-living/healthy-lifestyle/stress-management/lower-stress-how-does-stress-affect-the-body. Accessed March 18, 2021.
- Stress and your health. U.S. Department of Health & Human Services. https://www.womenshealth.gov/mental-health/good-mental-health/stress-and-your-health. Accessed March 18, 2021.
- AskMayoExpert. Stress management and resiliency (adult). Mayo Clinic. 2019.
- Seaward BL. Essentials of Managing Stress. 5th ed. Jones & Bartlett Learning; 2021.
- Seaward BL. Managing Stress: Skills for Self-Care, Personal Resiliency and Work-Life Balance in a Rapidly Changing World. 10th ed. Jones & Bartlett Learning; 2022.
- Olpin M, et al. Stress Management for Life. 5th ed. Cengage Learning; 2020.
- Hall-Flavin DK (expert opinion). Mayo Clinic. March 23, 2021.
Products and Services
- Newsletter: Mayo Clinic Health Letter — Digital Edition
- A Book: Mayo Clinic Guide to Home Remedies
- A Book: Mayo Clinic on High Blood Pressure
- A Book: Mayo Clinic Family Health Book, 5th Edition
- A Book: Live Younger Longer
- Headaches and stress
- Stress hair loss
- Stress symptoms
Mayo Clinic does not endorse companies or products. Advertising revenue supports our not-for-profit mission.
- Opportunities
Mayo Clinic Press
Check out these best-sellers and special offers on books and newsletters from Mayo Clinic Press .
- Mayo Clinic on Incontinence - Mayo Clinic Press Mayo Clinic on Incontinence
- The Essential Diabetes Book - Mayo Clinic Press The Essential Diabetes Book
- Mayo Clinic on Hearing and Balance - Mayo Clinic Press Mayo Clinic on Hearing and Balance
- FREE Mayo Clinic Diet Assessment - Mayo Clinic Press FREE Mayo Clinic Diet Assessment
- Mayo Clinic Health Letter - FREE book - Mayo Clinic Press Mayo Clinic Health Letter - FREE book
- Healthy Lifestyle
We’re transforming healthcare
Make a gift now and help create new and better solutions for more than 1.3 million patients who turn to Mayo Clinic each year.

IMAGES
VIDEO
COMMENTS
Sexual/Reproductive Health of Adolescents/Young People in Turkey-1. According to the 2000 census, the population of 10-19 and 20-24 age group is 20.7% and 9.8% of the general population, respectively in Turkey. The 2003 Turkish Demographic and Health Survey (TDHS) results show that women aged 15-24 constitute 15.8% of the female population.
Areas of work. Human Reproduction Programme. Monitoring and surveillance. Guidelines. Research. About us. We lead WHO's work on sexual and reproductive health across the life course. This work includes HRP, the UN's Special Programme of Research, Development and Research Training in Human Reproduction.
5,335 views. 4 articles. An innovative interdisciplinary journal which explores human reproductive health - from STIs to reproductive epidemiology - to advance universal access to sexual and reproductive health care.
of strategies for family planning and reproductive health services, and the health impacts of these strategies have been examined [27-30]. Nevertheless, there is a need for more impact evaluations of family planning interventions using randomized control trials, particularly in Sub-Saharan Africa where rigorous experimental evidence is scarce.
Sexual and reproductive health interventions to prevent adolescent pregnancy. Studies were included if any form of sexual and reproductive health education, counseling, and access to contraception was delivered to adolescents compared to no intervention or general health education. We identified 1,123 titles from the search conducted in all ...
Areas of work. SRH and HRP work collaboratively with partners across the world to shape global thinking on sexual and reproductive health by providing new ideas and insights. We work together to enable high-impact research, inform WHO norms and standards, support research capacity strengthening in low- and middle-income settings, and facilitate ...
Health Organization (WHO) defines reproductive health as a state of complete physical, mental and. social well-being, not just the absence of a reproductive disease or diseases. Reproductive ...
Reproductive Health and Research (SRH) conducted the global SRMNCAH policy survey in 2018-2019. This survey is the fifth such survey on maternal, newborn, child and adolescent health since 2009-2010, and it is the first to include sexual and reproductive health and to align with the SDGs and the Global Strategy for
To benchmark research on reproductive health and disease, we used the PubMed database to compare the number of articles published on seven reproductive organs and seven non-reproductive organs between 1966 and 2021 ().While the reproductive organs are not essential to postnatal life, we posit that the placenta and the uterus are as essential to fetal survival in utero as the lungs and the ...
The integration of human rights principles in sexual and reproductive health (SRH) research is often recognised to be of value. Good examples abound but lack of clarity persists as to what defines rights-inclusive SRH research. ... and agreement among key funding sources to mandate the integration of rights principles in research proposals they ...
Research that explores how community-based interventions for strengthening adolescent sexual reproductive health and rights (SRHR) can be integrated and sustained in community health systems, is, to the best of our knowledge, very scarce, if not absent. It is important to document mechanisms that shape integration process in order to improve health systems' responsiveness towards adolescents ...
Background An estimated 32 million women and girls of reproductive age living in emergency situations, all of whom require sexual and reproductive health (SRH) information and services. This systematic review assessed the effect of SRH interventions, including the Minimum Initial Service Package (MISP) on a range of health outcomes from the onset of emergencies.
Researchers at Emory University Center for Reproductive Health Research in the Southeast were approached as research partners with expertise in reproductive health data collection, analysis, and academic dissemination. ... Our proposal was funded and became the first community-led research study by the Society of Family Planning. Notably, all ...
Abstract. Adolescents have a high burden of sexual and repro ductive health problems and it is important to reach. out to youth groups through health promotion initiatives. A literature review was ...
Building upon formative, mixed-methods research conducted in sixteen countries between 2014 and 2016, the GEAS has collected baseline data from over 13,000 adolescents on five continents since 2017. Additional survey topics include sexual and reproductive health, mental health, body comfort, school retention and empowerment.
7 MARCH 2023 Request for Proposals: Sexual, Reproductive, and Maternal Health and Rights - 2023 3 | 11 • Be either a non-profit entity, research institution, for-profit social enterprise, or a team of such organizations. Individuals must apply through an institutional sponsor. Partnerships are strongly encouraged, but only the lead applicant will receive funds.
In many African countries, sexual and reproductive health (SRH) needs of young people / youth are often underserved and underestimated despite their demonstrated need and the urgency of these services [].Continental population remain high at approximately 1.2 billion with the highest number being youth aged 15-24 years, 226 million—19% of the global youth population—of whom live in sub ...
Specifically, this research proposal aims to accomplish the following objectives: Assess the existing reproductive health policies: Conduct a comprehensive review of national and international reproductive health policies, laws, and initiatives. Analyze the legal frameworks, funding mechanisms, and implementation strategies employed to promote reproductive health and rights.
Background Addressing the unmet need for modern contraception underpins the goal of all family planning and contraception programs. Contraceptive discontinuation among those in need of a method hinders the attainment of the fertility desires of women, which may result in unintended pregnancies. This paper presents experiences of contraceptive use, reasons for discontinuation, and future ...
In an important step for research on women's reproductive health, scientists have developed a cervix-on-a-chip, a lab model that replicates the structure and function of the human cervix. The team, led by researchers at Harvard Medical School, Boston Children's Hospital, the Wyss Institute for Biologically Inspired Engineering at Harvard ...
The Department of Population, Family and Reproductive Health is dedicated to improving health throughout the life course worldwide. We train research scientists, public health leaders, and health professionals to excel in careers related to a broad spectrum of population, family, and reproductive health issues.
Twenty, a non-government research organisation. One area of focus of this study is the emergence of sexual and lifestyle risk factors during adolescence, particularly risk behaviour, unwanted pregnancy, exposure to sexually transmitted infection etc (Birth to Twenty, 2005). The Reproductive Health Research Unit (RHRU) is another organisation
PMNCH Request for Proposals and Job Board Bulletin. PMNCH regularly advertises Requests for Proposals (RfPs) for various contracts related to work on maternal, newborn and child health. PMNCH often advertises Requests for proposals (RfPs) for activities and services, including consultancies.
Research-Proposal-Reproductive-Health (2).pptx - Free download as Powerpoint Presentation (.ppt / .pptx), PDF File (.pdf), Text File (.txt) or view presentation slides online. This study aims to determine the impact of the COVID-19 pandemic on the sexual and reproductive health of partner relationships in Tuguegarao City, Philippines.
The Ministry of Health, department of Sexual Reproductive Health; in partnership with Access For Action Uganda (ACFA Uganda) do present this proposal to promote sexual reproductive health for women and youth with disabilities in Wakiso district of Uganda 1 A/37/351/Add.1 and Corr.1, annex, sect. VIII, recommendation 1 (IV). 2 Resolution 48/96 ...
Systematic and rigorous inquiry allows us to discover the fundamental mechanisms and causes of disease and disparities. At our Office of Research (research@BSPH), we translate that knowledge to develop, evaluate, and disseminate treatment and prevention strategies and inform public health practice.Research along this entire spectrum represents a fundamental mission of the Johns Hopkins ...
The President's Fiscal Year (FY) 2025 Budget supports the Department of Health and Human Services' (HHS) mission to promote the health and well-being of all Americans. HHS proposes $130.7 billion in discretionary and $1.7 trillion in mandatory proposed budget authority for FY 2025.
NIH Releases Draft Policy Proposal to Promote Equal Access to Products Stemming from NIH-Owned Inventions. On May 22, the National Institutes of Health (NIH) released a draft policy proposal that would require organizations partnering with NIH through its licensing agreements to provide plans for patient access to those products.
Nevertheless, concerns have been raised regarding the effects of fasting on the reproductive health of women and men. For instance, some women are skeptical about starting intermittent fasting because they believe it may negatively affect levels of estrogen and other reproductive hormones, leading to menstrual cycle irregularities and fertility ...
Adrenaline makes the heart beat faster, causes blood pressure to go up and gives you more energy. Cortisol, the primary stress hormone, increases sugar, also called glucose, in the bloodstream, enhances the brain's use of glucose and increases the availability of substances in the body that repair tissues.